August 30, 2012 — Spinal cord stimulation (SCS) improves heart function and could become a novel treatment option for heart failure, according to research presented at the European Society of Cardiology Congress in Munich, Germany, by Hung-Fat Tse from Hong Kong.
August 30, 2012 — An ultra-fast, 320-detector computed tomography (CT) scanner can accurately sort out which people with chest pain need – or don’t need – an invasive procedure such as cardiac angioplasty or bypass surgery to restore blood flow to the heart, according to an international study.
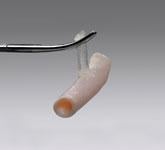
Vascular closure devices that use an active method to immediately seal the femoral access site can enable faster patient ambulation, reduce nursing time and speed discharge. However, one of the biggest issues interventionalists have with active vascular closure devices is the use of a permanent piece of hardware to stitch or clip the arteriotomy closed. Three companies now offer fully bioresorbable, active vascular closure devices, including a recent release earlier this year.
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...

The implementation of dedicated access site surveillance and educational programs, in tandem with pre-existent strategies to reduce access site complications, can reduce the overall incidence of femoral access-related bleeding events.

Cardiac computed tomography (CT), magnetic resonance imaging (MRI), echocardiography and nuclear myocardial perfusion imaging each offer advantages and disadvantages, and frequently at least two of these tests are required to get the full picture of a patient’s cardiac health. However, in this age of cutting healthcare costs, declining reimbursements and improving efficiency, it would be advantageous to have a single gold standard exam. Technology advances are now making that possibility.

The biggest issue in electrophysiology (EP) is the disparate nature of patient tests, reports, images and waveforms, and the need to aggregate all this data in one place to more efficiently care for patients with arrhythmias. This is especially true as the need to meet government-required meaningful use criteria for electronic medical records (EMRs) pushes forward and healthcare reform necessitates a deeper data integration.
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...

As vendors develop, and hospitals shop for, electrophysiology (EP) information and reporting systems, there are several considerations that will contribute to the efficiency and longevity of these systems.
August 29, 2012 — A new, daytime teleradiology company targeting urgent care, independent diagnostic testing facilities (IDTFs), mobile medical and physician offices is now serving clients in 10 states, is licensed in 20 and plans to expand with the growth of its customers. Teleradiology Specialists keeps overhead costs low and extends the savings to every customer while employing highly qualified radiologists who are available to read a wide variety of studies all day.
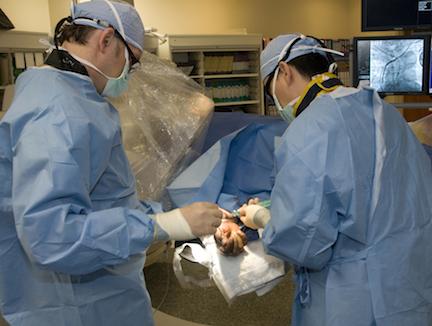
Recently, concerns have been raised that transradial (TR) cardiac catheterization significantly increases radiation exposure when compared with the conventional transfemoral (TF) approach. The current rapid uptake of TR in the United States is mainly due to its economic benefits and overall favorable patient outcomes. Perhaps most importantly, TR provides a measurable reduction in vascular complications, which translates to shorter time for ambulation and post-procedural stay. The reduction in bleeding with TR likely lowers mortality in patients with ST-elevation myocardial infarction (STEMI). One potential reason for the slow adoption of TR may be the perceived concern about increased radiation exposure.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...

Patients with 2C19 that is less active due to loss of gene copies are poor metabolizers of clopidogrel, opening patients to the possibility of a higher rate of mayocardial infacrtion (MI). There are assays for platelet function response (VerifyNow) and genetic tests to measure patient response, which could help determine if generic clopdogrel or more expensive agents should be used.

The standard-of-care for dual antiplatelet therapy (DAPT) for more than a decade has been clopidogrel (Plavix) plus aspirin. This standard is now being challenged by new agents — ticagrelor (Brilinta) and prasugrel (Effient). While the new agents are more effective at preventing platelet aggregation than clopidogrel, each has unique characteristics. Plavix went generic earlier this year, which may also impact decisions about which therapies to use.
August 28, 2012 — Toshiba America Medical Systems Inc. has received U.S. Food and Drug Administration (FDA) clearance on its new Vantage Titan 1.5T series, which features the 8-, 16- and 32-channel MR (magnetic resonance) systems.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
August 28, 2012 — The International Contrast Ultrasound Society (ICUS) applauded the U.S. Food and Drug Administration’s (FDA) decision to modify the U.S. product label for several ultrasound contrast agents used to improve the accuracy of radiation-free ultrasound scans.
August 28, 2012 — GE Healthcare announced important changes to the U.S. product label for Optison (perflutren protein-type A microspheres injectable suspension, USP), a contrast agent that may improve visualization of the left ventricular border, an area of the heart that is critical to see in order to diagnose certain heart diseases such as hypertrophic cardiomyopathy.
August 28, 2012 — NDS Surgical Imaging (NDSsi) is now shipping its new Dome GX4MP radiology display, a 30-inch widescreen model offering multimodality viewing in both color and grayscale.


 August 30, 2012
August 30, 2012








