Years ago, I owned a computer that ran a spreadsheet program called Lotus 1-2-3. After about a year, I needed to perform ...
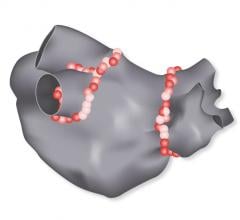
Biosense Webster Inc. recently received approval from the U.S. Food and Drug Administration (FDA) for its Visitag Surpoint External Processing Unit, and enrollment has begun in its post-market approval study.
The U.S. Food and Drug Administration (FDA) has cleared the Magnetom Sola, a 1.5 Tesla magnetic resonance imaging (MRI) scanner from Siemens Healthineers that brings Siemens’ BioMatrix technology to the 1.5T market. This technology addresses patient anatomical and physiological differences, as well as differences in how users set up and conduct MRI exams, to increase productivity and decrease rescans for improved efficiency and patient satisfaction.
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
Tracking just seven factors of heart attack patients when they are first admitted to the hospital can help flag those most at risk for 30-day readmission, researchers from UT Southwestern found.
The U.S. Food and Drug Administration (FDA) granted market clearance for FibriCheck, a Belgian medical smartphone application for the detection of heart rhythm disorders. The approval makes FibriCheck the first-FDA approved app for heart rhythm disorders by using only an optical signal originating from a non-medical device such as a smartphone.
Endologix Inc. received notice that the U.S. Food and Drug Administration (FDA) has classified a voluntary recall action that Endologix took in July of this year as a Class I recall. The July recall involved Endologix’s issuance of a Safety Notice to healthcare professionals (HCPs) using the AFX Endovascular AAA System.
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...

“I don’t trust anything that’s being portrayed in the marketing brochures and will believe it only when the solution is ...
Veryan Medical Ltd has received Premarket Approval (PMA) for the BioMimics 3D Vascular Stent System from the U.S. Food & Drug Administration (FDA). The device is approved for the treatment of symptomatic de novo or restenotic lesions in the native superficial femoral artery and/or proximal popliteal artery.
Teleflex Inc. announced the acquisition of Essential Medical Inc. Based in Exton, Pa., Essential Medical is a privately-held medical device company that has developed and commercialized the CE Marked Manta Vascular Closure Device specifically designed for closure of large bore arteriotomies following procedures utilizing devices or sheaths ranging in size from 10F to 18F (with maximum outer diameters up to 25F).
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
The Ohio State University Wexner Medical Center is first in the country to test a new medical device designed to help patients with advanced heart failure. Cardiologists here performed the first two implants and the patients went home the next day.
October 4, 2018 — The late-breaking FAST-FFR Trial demonstrated that the sensitivity and specificity of the CathWorks ...
Fujifilm SonoSite Inc. announced its entry into the medical informatics space with the launch of a robust point-of-care workflow solution, SonoSite Synchronicity. The software is designed specifically for point-of-care ultrasound to streamline and standardize clinical workflow while delivering administrative efficiencies with flexible credentialing tools.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
October 4, 2018 — Pulmonary artery denervation (PADN) is associated with significant improvements in hemodynamic and ...
Three-dimensional printing company Biomodex, which develops organ twins for advanced physician training, is expanding into the interventional cardiology space with the launch of the Left Atrial Appendage Closure Solution (LAACS). Unveiled at the 2018 Transcatheter Cardiovascular Therapeutics (TCT) conference, Sept. 21-25 in San Diego, the LAACS allows physicians to train on an ultra-realistic, multi-material, 3-D-printed heart.
October 4, 2018 – Investigators unveiled clinical data from the independent BIONYX and SORT OUT IX all-comers trials ...

 October 10, 2018
October 10, 2018