April 18, 2018 — The U.S. Food and Drug Administration (FDA) has cleared the Somatom go.All and Somatom go.Top computed ...
Northwestern Medicine has purchased several smaller Chicago suburban hospitals in the past few years to expand its ...
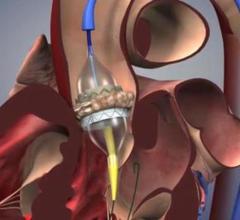
April 17, 2018 — The U.S. Food and Drug Administration (FDA) announced market approval for the Abbott Perclose ProGlide ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
Philips Healthcare last week issued a proactive advisory warning to its iSite and IntelliSpace picture archiving and communication system (PACS) customers of potential security vulnerabilities in the products. The company cautioned that while it has received no reports of patient harm, the vulnerabilities in question could impact or potentially compromise patient confidentiality, system integrity and/or system availability.
The skin presents a formidable barrier to life-saving defibrillators, but a team of students from Rice University believes it has found a way around that problem.

Over the last decade, there have been considerable developments in procedural techniques and technology facilitating the ...
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
Medtronic plc announced U.S. Food and Drug Administration (FDA) approval to begin an investigational device exemption (IDE) pivotal trial to evaluate the Symplicity Spyral renal denervation system in patients with high blood pressure (hypertension). Renal denervation is a minimally invasive procedure intended to regulate the activity of nerves that lead to and from the kidney, which plays an important role in managing blood pressure. The SPYRAL HTN Pivotal Trial is part of the broader SPYRAL HTN Global Clinical Program, a multi-phased clinical study strategy aimed to establish the safety and efficacy of renal denervation to lower blood pressure.
Canon Medical Systems USA Inc. this week received U.S. Food and Drug Administration (FDA) clearance for the Aquilion Precision system, which it calls the world’s first Ultra-High Resolution computed tomography (UHR CT) system. The system can resolve anatomy as small as 150 microns and is designed to provide more than twice the resolution, typically seen only in cath labs. Containing an all-new detector as well as tube, gantry and reconstruction technologies, the system may make it possible to help expand visualization of disease thanks to new features that offer improved image detail.
April 13, 2018 — Medical advice about implanted cardiac defibrillators obtained via an online message board appears to ...
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
Biotronik U.S. and Aziyo announced a strategic agreement allowing Biotronik to distribute Aziyo's CanGaroo extracellular matrix (ECM) cardiovascular implantable electronic device (CIED) envelopes in the United States. BioEnvelope will be available from Biotronik starting in April 2018.
French medical technology company CorWave announced it has been awarded almost $3.5 million in financing for its R&D program NovaPulse in the Silver Economy category. This is part of phase 2 of France’s Concours Mondial d'Innovation or Worldwide Innovation Challenge.
Women who give birth to infants with congenital heart defects may have an increased risk of cardiovascular hospitalizations later in life, according to new research in the American Heart Association’s journal Circulation.
Change Healthcare Cardiology Hemodynamics is an integrated hemodynamic monitoring system for monitoring vital signs and ...
Medtronic plc announced one-year results from the CRYO4PERSISTENT AF study of ablation with the Arctic Front Advance Cryoballoon to isolate the pulmonary veins in patients with symptomatic persistent atrial fibrillation (AF). The Arctic Front Advance Cyroablation System is not approved for treating persistent AF in the United States.
The biggest workplace concern for interventional cardiologists and cath lab staff is their daily exposure to ionizing ...
Abbott today announced the initiation of a clinical trial evaluating long-term outcomes of patients who undergo stent implantation guided by optical coherence tomography (OCT) compared to angiography. The trial (ILUMIEN IV) is the first large-scale randomized global study using Abbott's OCT imaging in patients with high-risk, complex coronary artery disease. Patients in the study will be randomized to either OCT-guided or traditional angiography to guide placement of one or more Xience everolimus-eluting coronary stents.

 April 18, 2018
April 18, 2018