September 30, 2008 – Czech Republic-based Tryton Medical Inc. said today it launched a novel Side Branch Stent System in Europe.
Tryton’s Side Branch Stent System is designed to offer a dedicated strategy for treating atherosclerotic lesions at the site of a bifurcation. The system will be available first in the Netherlands, with additional European countries to follow in the coming months. TOP Medical has partnered with Tryton to distribute the device in the Netherlands.
Cardiovascular disease causes nearly half of all deaths in Europe, claiming 4.3 million lives annually. Approximately 20 percent of patients treated for coronary artery disease are treated for bifurcated lesions.
Bifurcation lesions have presented a challenge for cardiologists since the earliest days of angioplasty. Current approaches to treating these lesions have significant limitations. As a result, the side branch is often left unstented, leaving it vulnerable to higher rates of restenosis, the re-narrowing of the stented vessel following implantation.
“The Tryton Side Branch Stent System, which has demonstrated excellent initial clinical results, offers an innovative solution to a persistent problem in the interventional treatment of patients with complex coronary lesions,” said professor Antonio L. Bartorelli of the Centro Cardiologico Monzino at the University of Milan, who attended the European Bifurcation Club meeting in Prague where the launch was announced. “I am pleased that this highly deliverable side-branch stent is now available as a treatment option for these difficult-to-treat cases.”
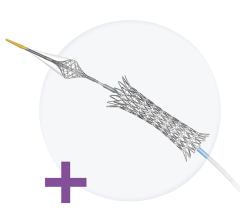
The Tryton Side Branch Stent System is used in conjunction with a conventional drug-eluting stent.
The Side Branch Stent System is designed to offer a dedicated strategy for treating atherosclerotic lesions in the side branch at the site of a bifurcation. These areas of the vascular system are a common location for plaque and are particularly challenging to treat with currently available stent systems. About 20 percent of patients treated for coronary artery disease are treated for bifurcated lesions.
Tryton’s highly deliverable cobalt chromium stent is deployed in the side branch artery using a standard single-wire balloon-expandable stent delivery system. A conventional drug eluting stent is then placed in the main vessel.
The Tryton Side Branch Stent System demonstrated excellent six-month clinical results in a first-in-man study of the system in 30 patients, with no restenosis occurring in the side-branch artery. The stent system has received CE Mark approval in Europe and is not approved in the U.S.
For more information: www.trytonmedical.com


 November 24, 2025
November 24, 2025