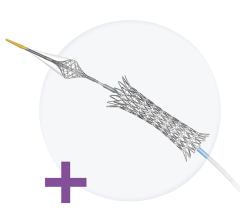
February 13, 2008 - Nfocus Neuromedical Inc. announced that the company has received CE mark approval for the CardioVasc Stent-Graft and Delivery System, a minimally invasive device for treating patients with Saphenous Vein Graft Lesions (SVGs) from a previous coronary bypass, Vessel Perforations and Coronary Artery Aneurysms.
Saphenous vein graft (SVG) disease has been a problem facing the cardiologist since the early days of coronary artery bypass graft (CABG) surgery said the company. In CABG surgery, a segment of the patient's own leg vein (the saphenous vein) may be removed and used to create an artificial conduit to bypass areas of blood flow blockage (also known as stenosis or plaque) that have built up in the patient's coronary artery. After CABG surgery, this SVG bypass itself may become blocked over time by the same type of disease that affected the patient's original coronary artery. The result is a need to reopen the diseased SVG vessel and restore flow. This second treatment again can be yet another surgery, or alternatively, the SVG may be treated via angioplasty and stenting procedures using less invasive endovascular (catheter) techniques. The CardioVasc Stent-Graft device is a permanent implant delivered via a less-invasive catheter procedure for patients that have a diseased SVG from a previous CABG surgery.
For more information: www.nfocusneuro.com


 November 24, 2025
November 24, 2025