Christine Albert, M.D., MPH, professor, chair of the Department of Cardiology and the Lee and Harold Kapelovitz ...
EP Lab
This channel includes news and new technology innovations for cardiac electrophysiology (EP) systems, techniques and devices using in EP labs. This includes implantable EP devices, pacemakers, implantable cardioverter defibrillators (ICD), cardiac resychronization therapy (CRT), ablation technologies, left atrial appendage (LAA) occlusion, atrial fibrilation (AF) and Holter monitors.

November 18, 2021 — The U.S. Food and Drug Administration (FDA) is reminding providers about the risk of major ...
November 17, 2021 — People with atrial fibrillation (AFib) who underwent individualized testing to discover triggers for ...
When the patients of Michael Boler, M.D. need cardiac monitoring, the Holter monitor is no longer his first choice. “The ...
November 16, 2021 — Acutus Medical Inc. (Acutus), an arrhythmia management company focused on improving the way cardiac ...
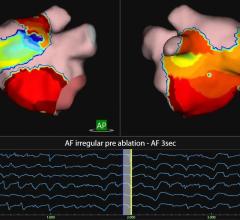
November 15, 2021 — Vektor Medical Inc. announced U.S. Food and Drug Administration (FDA) 510(k) clearance for its ...
November 12, 2021 — Orchestra BioMed Inc. announced multiple presentations of long-term clinical results and ISH ...
Apple created a stir when it announced in 2018 that its Apple Watch Series 4 was the first consumer health and fitness ...
November 2, 2021 — Cardionomic Inc. announce initial U.S. enrollment in its global Cardiac Pulmonary Nerve Stimulation ...

November 1, 2021 — Here is the list of the most popular content on the Diagnostic and Interventional Cardiology (DAIC) ...
With 1.2 to 1.4 million new electrophysiology (EP) devices being prescribed to patients around the world each year ...
Washington Health System (WHS) provides healthcare services at more than 40 offsite locations across three counties in ...
October 28, 2021 — CathVision, a medical technology company developing electrophysiology (EP) solutions in EP recording ...
October 27, 2021 — Impulse Dynamics announced the U.S. Food and Drug Administration approved a modification of labeling ...
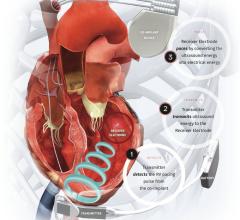
October 27, 2021 — EBR Systems Inc., developer of the world’s first wireless cardiac pacing system for heart failure, ...
Apple created a stir when it announced in 2018 that its Apple Watch Series 4 was the first consumer health and fitness ...
An example of the Acutus Medical AcQMap High Resolution Imaging and Mapping System to guide electrophysiology (EP) cardi ...
October 6, 2021 — Boston Scientific Corp. announced it entered into a definitive agreement to acquire Baylis Medical ...
October 6, 2021 – Biosense Webster announced the first cases using a radiofrequency balloon ablation catheter were ...


 November 22, 2021
November 22, 2021






![Figures from an initial study on the Cardionomic CPNS technology in a poster presentation at the Heart Rhythm Society (HRS) 2021 meeting.[1] Starting at top left, an angiographic view of the atrial transseptal access for a left ventricular septal ablation. Procedural intra-cardiac echo (ICE) showing ablation catheter positioning. A 3D electro-anatomic map of the LV septum. The final graphs show baseline and post-procedure LVOT pressure readings demonstrating a decreased gradient.](/sites/default/files/styles/content_feed_medium/public/Cardionomic_Cardiac_Pulmonary_Nerve_Stimulation_CPNS_for_HF.jpg?itok=ecrU9hmQ)