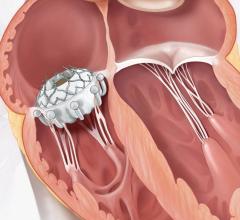
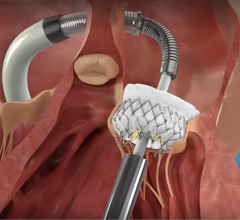
The Baylis VersaCross RF transseptal crossing catheter is designed to puncture the septum without a needle to allow access to the left side of the heart for mitral valve and left atrial appendage occlusion procedures. Boston Scientific to Acquire Baylis Medical for $1.75 billion.
October 6, 2021 — Boston Scientific Corp. announced it entered into a definitive agreement to acquire Baylis Medical Company Inc. for an upfront payment of $1.75 billion, subject to closing adjustments. The acquisition will expand the Boston Scientific electrophysiology (EP) and structural heart product portfolios to include the radiofrequency (RF) NRG and VersaCross transseptal puncture platforms, as well as a family of guidewires, sheaths and dilators used to support left heart access.
These platforms have advanced transseptal puncture and are clinically proven to enhance safety, efficacy and efficiency when crossing the atrial septum to deliver therapies in the left side of the heart, such as atrial fibrillation ablation, left atrial appendage closure (LAAC) and mitral valve interventions.[1,2,3] Baylis is expected to generate net sales approaching $200 million in 2022, having achieved double-digit, year-over-year sales growth during each of the past five years.
"The talented and innovative Baylis Medical Company team, combined with these transseptal platforms, will enhance our efforts to improve procedural efficiencies with physician tools designed to make left atrial access safer and more predictable, with a focus on patient outcomes," said Mike Mahoney, chairman and chief executive officer, Boston Scientific. "A leader in many of the fastest growing markets in our industry, we believe that Baylis Medical Company will add meaningful revenue, operating income, and new research and development capabilities across multiple Boston Scientific businesses, while complementing existing offerings within our electrophysiology and structural heart portfolios."
Physicians have traditionally relied on a mechanical needle to pass through the septum and access the left side of the heart, which can present safety concerns and placement challenges based on varying patient anatomy. Rather than relying solely on mechanical force, the Baylis Medical platforms facilitate predictable and safe transseptal access by using RF energy – a method shown to increase efficiency, and improve the safety and efficacy of transseptal puncture during left heart procedures.[1,2,3]
The new VersaCross platform further streamlines transseptal crossing procedures and therapy delivery by offering the same benefits while eliminating potential wire and sheath exchanges, which may help mitigate risks during procedures.
"As a leading innovator in left heart access solutions, we develop advancements that help physicians deliver critical, high-precision therapies, which raise the standard of care for patients," said Kris Shah, president of Baylis Medical. "We look forward to making these life-changing technologies available to more patients across the globe through the significant commercial reach of Boston Scientific."
Baylis Medical received U.S. Food and Drug Administration (FDA) 510(k) clearance for the NRG platform in 2008 and the technology has since been used in more than 1 million procedures. The company received FDA 510(k) clearance for the VersaCross platform in 2020. These platforms are complemented by the company's family of guidewires, sheaths and dilators, which are designed for use in left-sided diagnostic, ablation, mitral and LAAC procedures.
The transaction is anticipated to close in the first quarter of 2022, subject to customary closing conditions.
References:
2. Winkle et al. The use of a radiofrequency needle improves the safety and efficacy of transseptal puncture for atrial fibrillation ablation. Heart Rhythm, Volume 8, Issue 9, 2011. DOI 10.1016/j.hrthm.2011.04.032
3. Inohara et al. VersaCross radiofrequency system reduces time to left atrial access versus conventional mechanical needle. J Interv Card Electrophys, 2021; in press (DOI: 10.1007/s10840-020-00931-7)


 July 08, 2024
July 08, 2024