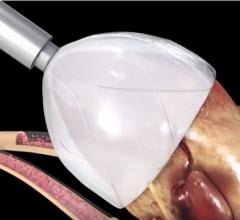
December 29, 2017 — Vivasure Medical announced in October the successful enrollment of the first patient in the Frontier ...
Cath Lab
The catheterization lab channel includes content related to interventional technologies for coronary and peripheral artery disease (PAD). Other cath lab transcatheter device technologies covered on this page included percutaneous treatment for stroke, venous interventions, heart valves, hypertension, heart failure and percutenous coronary interventions (PCI).
There have been many innovative new technologies highlighted at the medical conferences I attended in 2017. Each year, I ...

The Diagnostic and Interventional Cardiology (DAIC) website had another record year with 1.4 million page views in 2017 ...
Adjustable parameters on the ACIST® CVI® Contrast Delivery System include, flow rate, volume, pressure limit, rise time ...
December 28, 2017 — Corindus Vascular Robotics Inc. announced recently that Houston Methodist DeBakey Heart & Vascular ...
December 28, 2017 — HeartFlow Inc. announced that Health Care Service Corp. (HCSC), which operates Blue Cross and Blue ...
December 22, 2017 — Essential Medical announced the completion of enrollment in the U.S. pivotal investigational device ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...
December 19, 2017 — A team of researchers at the National Institute for Health Research (NIHR) Leicester Biomedical ...
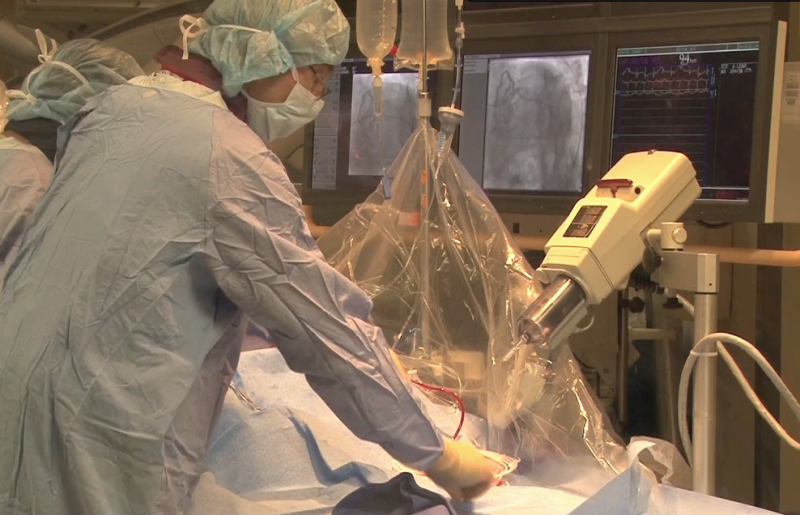
As payers and other healthcare entities look to better manage costs, especially in the acute care setting, it is ...
December 14, 2017 — Vasorum Ltd, the developer and manufacturer of the novel Celt ACD vascular closure device, has added ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
December 14, 2017 — The U.S. Food and Drug Administration (FDA) announced clearance for NuMed’s Cheatham Platinum (CP) S ...
December 13, 2017 — Cordis, a Cardinal Health company, and Medinol recently announced U.S. Food and Drug Administration ...
December 11, 2017 — CorInnova Inc. recently announced it was awarded second prize in the “2017 InnoSTARS” life science ...
During cardiology fellowship, my institution had a hand manifold system. I found it difficult to use at times, and it ...
December 7, 2017 — Edwards Lifesciences Corp. announced the acquisition of Harpoon Medical Inc., a privately held ...
December 6, 2017 — French company Robocath, which designs and develops robotic solutions for the treatment of vascular ...

Here is the list of the most popular articles and videos on the Diagnostic and Interventional Cardiology (DAIC) magazine ...


 December 29, 2017
December 29, 2017