Agfa began its global release of ICISTM View 3.0, the medical images and results viewer for the comprehensive ICIS enterprise imaging solution. The ICIS patient imaging data platform is a fundamental part of the company’s global e-health strategy. Building on its leadership in enterprise imaging management and web-enabled technology, Agfa HealthCare’s ICIS View 3.0 allows clinicians, specialists, and all other stakeholders to access all patient imaging data from any PACS or VNA, using a single viewer, to support continuity and productivity of patient care. This cost- effective solution leverages the hospital’s existing investment in technology, and provides access to the most current and relevant patient imaging data directly from the source.
St. Jude Medical Inc. announced U.S. Food and Drug Administration (FDA) approval to begin the EnligHTN IV Renal Denervation Study, the first U.S. trial using the EnligHTN Multi-Electrode Renal Denervation System to treat patients with drug-resistant high blood pressure.
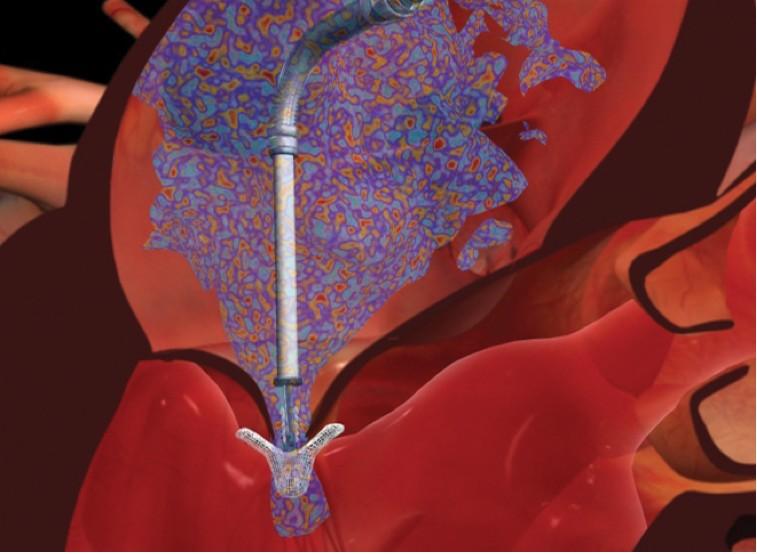
Abbott has announced publication of positive outcomes from two European post-approval studies of the catheter-based MitraClip therapy for the treatment of mitral regurgitation (MR). Results from ACCESS-EU, a European prospective study that enrolled 567 patients at 14 sites, have been published in the Journal of the American College of Cardiology. In addition, findings of the investigator-sponsored German TRAnscatheter Mitral Valve Interventions (TRAMI) registry, which enrolled 1,064 patients at 20 German sites, were recently published in EuroIntervention.
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
Tryton Medical Inc. announced that the Nordic-Baltic Bifurcation Study Group will investigate the Tryton Side Branch Stent. The trial is a prospective, controlled, randomized, multicenter clinical study examining the role of final kissing balloon inflations in patient outcomes. A total of 150 patients will receive the Tryton Side Branch Stent with a drug-eluting stent, with evaluation by intravascular optical coherence tomography (OCT) imaging technology.
Mirada Medical collaborators has presented new positron emission tomography/magnetic resonance imaging (PET/MRI) research. The software supports both the hardware hybrid scanners and provides validated deformable registration for software based PET/CT (computed tomorgraphy)/MRI.
MIM Software Inc. has introduced a new version of MIM Encore that provides a single platform for viewing positron emission tomography (PET), computed tomography (CT), magnetic resonance imaging (MRI) and nuclear medicine exams. Users will benefit from having all of the imaging data they need available on one workstation. Additionally, integrated quantitative analysis solutions are available for both cardiac and neuro PET and single photon emission computed tomography (SPECT) providing an all-in-one solution for Nuclear Medicine departments.
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
Siemens Healthcare has introduced the world’s first xSPECT system — combines the high sensitivity of single-photon emission computed tomography (SPECT) with the high specificity of computed tomography (CT), completely integrating the data from the two modalities, to generate high resolution and quantitative images. Meanwhile, Biograph mCT Flow is a new positron emission tomography/computed tomography (PET/CT) system that overcomes the limitations of conventional bed-based PET/CT with FlowMotion. This new technology moves the patient smoothly through the system’s gantry, while continuously acquiring PET data.
UltraSPECT announced the installation of its proprietary Wide Beam Reconstruction (WBR) software at nearly 10 healthcare facilities. The installations came as a result of the recent agreement between UltraSPECT and radiopharmaceutical provider PharmaLogic, under which PharmaLogic offers the WBR software as part of its patient-centered approach.
Siemens Healthcare has introduced Biograph mCT Flow, a positron emission tomography/computed tomography (PET/CT) system that, for the first time ever, overcomes the limitations of conventional bed-based PET/CT with FlowMotion, a revolutionary new technology that moves the patient smoothly through the system’s gantry, while continuously acquiring PET data. Biograph mCT Flow with FlowMotion takes routine image quality to a new level by enabling imaging protocols based on the organ’s need.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
The phrase “doing more with less” is becoming more prevalent in tightening economic environments, and the operating room (OR) is one example of where hospitals are looking to maximize savings. Mobile C-arm units are important for maximizing space in the OR and can provide the image quality needed to visualize anatomy and devices during complex surgical procedures. The latest advances in mobile C-arms aim to provide users with the flexibility and ergonomics that can support accomplishing more in the OR with less.
New long-term data from the DIVERGE study, presented at EuroPCR 2013 by Principal Investigator Dr. Stefan Verheye, Antwerp Cardiovascular Centre, ZNA Middelheim Hospital, Belgium, has shown that the use of the Axxess drug-eluting stent (DES) for the treatment of complex coronary bifurcation lesions resulted in low levels of both MACE and VLST over a five-year period. Axxess is now the only dedicated bifurcation stent with a substantial body of supporting data out to five years.
Between June and October, 2013, healthcare providers at hospitals and medical centers across the United States and Canada can experience GE Healthcare’s entire line of Centricity* Imaging technologies when the Centricity Imaging IT Tour visits locations in over 30 major markets.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
eHealth Technologies’ eHealthViewer ZF is a zero footprint viewer that enables clinicians, with any secure browser, to instantly access their patients’ medical images, in diagnostic-quality, right from within their patients’ medical record. The latest enhancement to the eHealthViewer ZF is Image Collaboration. Whether through consultation with a remote specialist or grand rounds with the patients’ clinical care team, the eHealthViewer ZF collaboration tool delivers real-time, fluid image review and interaction for healthcare professionals. Using this singular feature, multiple users at different locations, can interactively view and manipulate radiology and cardiology images with full access to the eHealthViewer ZF advanced toolset.
Just a few years ago, integrated positron emission tomography and magnetic resonance (PET/MR) imaging was found only in research institutes, but little by little the technology has expanded into clinical practice. This is especially true for cardiac indications, for which the highly sensitive soft tissue contrast of MR and the functional and metabolic imaging of PET are particularly valuable. New research proves the value of PET/MR compared to PET/computed tomography (CT) in cardiac applications, say researchers at the Society of Nuclear Medicine and Molecular Imaging’s 2013 Annual Meeting.

Direct Flow Medical Inc. announced that it has received approval from the U.S. Food and Drug Administration (FDA) for an Investigational Device Exemption (IDE) to begin the SALUS feasibility trial of the Direct Flow Medical Transcatheter Aortic Heart Valve System. The device encompasses a transcatheter aortic heart valve with a metal-free frame and flexible, low-profile delivery system that virtually eliminates aortic regurgitation. It is designed to improve the long term survivability of patients by resolving the clinical issues associated with current commercial valves.


 June 13, 2013
June 13, 2013









