Digisonics and Medis have partnered to provide a single system solution for image analysis and structured reporting of pediatric and adult cardiovascular magnetic resonance (MR) studies.
A University of Alabama at Birmingham surgical team has performed the first surgery using a virtual augmented reality technology called VIPAAR (Virtual Interactive Presence in Augmented Reality) in conjunction with Google Glass, a wearable computer with an optical head-mounted display.

Comprised of the cardiologist and the heart surgeon, the heart team collaboratively assesses and determines the best course of treatment for cardiac patients. ECHI has a large and successful surgical and percutaneous valve program built on the heart team concept. This approach is also used for treating patients with coronary artery disease by applying the use of fractional flow reserve (FFR).
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
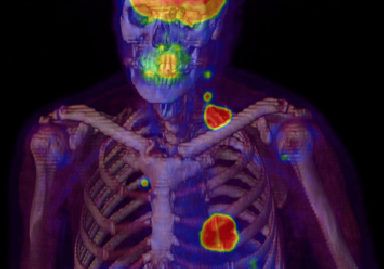
A report released by IMV Medical Information Division found that providers of nuclear medicine (NM) services are consolidating as procedure volume dropped in the subspecialty from 2008 to 2012.
The National Institutes of Health announced that a network of 25 regional stroke centers working with nearby satellite facilities will span the country, have teams of researchers representing every medical specialty needed for stroke care and will address the three prongs of stroke research: prevention, treatment and recovery.
AtriCure Inc., an atrial fibrillation (AF) medical device provider, and Endoscopic Technologies Inc, doing business as “Estech,” announced that they have entered into a definitive merger agreement under which AtriCure has agreed to acquire Estech.
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...

A study shows that coronary artery calcium (CAC) screening, an assessment tool that is not currently recommended for people considered at low risk, should play a more prominent role in helping determine a person’s risk for heart attack and heart disease-related death, as well as the need for angioplasty or bypass surgery.
Clinical trials for a promising new class of cholesterol-lowering medications, a long-awaited repair of the Medicare formula for paying physicians, the Affordable Care Act, new technology and the implementation of new prevention guidelines are among the top developments in cardiology anticipated for 2014.

It was not until we began planning that we realized just how involved TAVR programs are — they require much more work than we originally anticipated. At Terrebonne General Medical Center (TGMC), our TAVR program started as part of a research project in our structural heart department. It was an opportunity to offer advanced patient care and determine the effectiveness of TAVR on patients experiencing severe aortic stenosis, who were deemed inoperable by two cardiac surgeons.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
Intact Vascular Inc., a developer of medical devices for minimally invasive peripheral vascular procedures, announced the start of enrollment in the Tack Optimized Balloon Angioplasty Below the Knee (TOBA-BTK) study.
Mobile Aspects announced that it has been awarded the patent for its iRISupply radiofrequency identification (RFID) system that supports the storage and management of high-value medical devices and surgical products used in specialty areas of a hospital.
Atrial fibrillation (AF), long considered the most common condition leading to an irregular heartbeat, is a growing and serious global health problem, according to the first study ever to estimate the condition’s worldwide prevalence, death rates and societal costs.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
Careful documentation of patient selection and procedure appropriateness are critical — yet underutilized — steps in ensuring high-quality clinical care for percutaneous coronary intervention (PCI).
AliveCor announced Android compatibility for its AliveInsights analysis service, which launched for iOS (iPhone) users of the AliveCor Heart Monitor Nov. 18, 2013.
Following public comment received in the fall of 2013, The Joint Commission has released new accreditation standards for diagnostic imaging services. Nearly all of these new standard Elements of Performance (EP) are slated to become effective in a half-year.

 December 30, 2013
December 30, 2013









