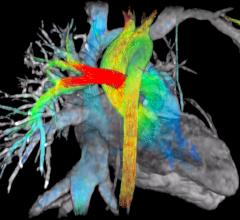
Arterys Inc. launched the Arterys System, an intelligence platform designed to enhance medical imaging, at the 101st Scientific Assembly and Annual Meeting of the Radiological Society of North America (RSNA), Nov. 29-Dec. 3 in Chicago.
The eighth Image Wisely Radiation Safety Case — C-arm Based Cone Beam CT in Interventional Radiology — is now available to help radiologists, imaging technologists and medical physicists assess their understanding of important radiation safety concepts — including dose monitoring and optimization.
Researchers from the University of Toronto have found that a specific cell type plays a key role in maintaining healthy arteries after inflammation.
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
At Ahuja Medical Center, University Hospitals (UH) cardiologists recently implanted the Boston Scientific Emblem Subcutaneous Implantable Cardioverter-Defibrillator (S-ICD) system, the first such procedure done at this hospital.
Biotronik announced the first patient enrollments to the B3 clinical trial. The study will evaluate the potential clinical benefit of the physiologic rate response sensor Closed Loop Stimulation (CLS) in atrial fibrillation (AF) patients.
December 3, 2015 — Clinically viable model-based iterative reconstruction is now a reality with the launch of Toshiba ...
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
Feeling high levels of distress, fear and hostility prior to undergoing an angioplasty or other interventional radiology procedure may lead to a poor outcome, according to new research presented today at the annual meeting of the Radiological Society of North America (RSNA).
Philips announced IntelliSpace Portal 8.0, the latest edition of its advanced data sharing, analytics and visualization platform that helps radiologists detect, diagnose and follow up on treatment of diseases.

December 2, 2015 — Researchers in the Netherlands studying thousands of healthy adults have found a connection between ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...

December 2, 2015 — Researchers using modern imaging techniques on hearts more than 400 years old found at an ...
December 1, 2015 — Philips introduced the DoseWise Portal 2.0, a comprehensive radiation dose management software ...
December 1, 2015 — An interventional radiology technique shows promise for helping morbidly obese patients lose weight ...
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
December 1, 2015 — Athletes who engage in the extreme sport of free diving, descending hundreds of feet below the ...
Frost & Sullivan presented the 2015 European Award for Technology Leadership to Ebit, the Esaote Group’s healthcare IT company focused on developing and selling RIS/CVIS/PACS software for integrated workflows in hospital enterprise processes.
Medic Vision Imaging Solutions Ltd. announced the introduction of SafeCT-29. Designed to be computed tomography (CT)-vendor neutral and compatible with the various makes and models of CT systems, SafeCT-29 helps meeting the NEMA XR-29 (Smart Dose) standard and avoiding Centers for Medicare and Medicaid Services (CMS) penalties.

 December 09, 2015
December 09, 2015














