Philips recently announced that it acquired VitalHealth, a provider of cloud-based population health management solutions for the delivery of personalized care outside of the hospital, for example, in regional care networks. Headquartered in the Netherlands, VitalHealth’s products and services are being used by more than 100 healthcare networks in various countries including the United States, India, China, Sweden, Germany, Belgium and the Netherlands. The company was founded in 2006 by Mayo Clinic (U.S.) and Noaber Foundation (the Netherlands) and employs approximately 200 employees. Financial details of the transaction were not disclosed.
December 22, 2017 — To help streamline treatment of patients with heart failure and ensure the best possible patient ...
Essential Medical announced the completion of enrollment in the U.S. pivotal investigational device exemption (IDE) trial of the Manta large bore vascular closure device. The company said it has enrolled 341 patients at 21 different sites with 43 investigators in 10 months. Essential Medical President and CEO Greg Walters said the company expects to file a premarket approval (PMA) submission with the U.S. Food and Drug Administration (FDA) by the end of the first quarter in 2018.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
December 21, 2017 — Bay Labs, a medical technology company applying artificial intelligence (AI) to cardiovascular ...
The Centers for Medicare & Medicaid Services (CMS) last week finalized the cancellation of the mandatory hip fracture and cardiac bundled payment models that were to be operated by the CMS Innovation Center and implemented changes to the Comprehensive Care for Joint Replacement (CJR) Model. These changes will offer greater flexibility and choice for hospitals in providing care to Medicare patients.
Philips recently announced agreements with 3D Systems and Stratasys, two global leaders in the 3-D printing industry, to help progress patient care and improve the clinician experience. Advanced 3-D modeling provides radiologists with additional views to help strengthen anatomical knowledge which could enhance clinical impact in reviewing complex, multi-disciplinary cases.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
A team of researchers at the National Institute for Health Research (NIHR) Leicester Biomedical Research Centre, U.K., has been awarded €6.5 million from the Horizon 2020 European Commission fund. The team — a partnership between Leicester’s Hospitals, the University of Leicester and Loughborough University, and led by clinical scientist consultant Prof. Anthony Gershlick from the university’s Department of Cardiovascular Sciences — will investigate the impact of using extracorporeal membrane oxygenation (ECMO) on the outcomes of patients who have had a severe heart attack.
ERT recently announced it has acquired iCardiac Technologies, a provider of centralized cardiac safety and respiratory solutions that accelerate clinical research. Financial terms of the transaction were not disclosed.
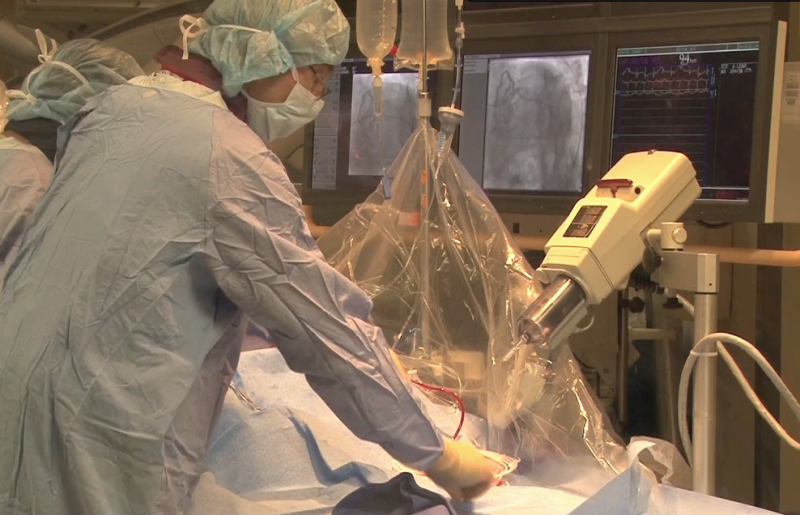
As payers and other healthcare entities look to better manage costs, especially in the acute care setting, it is ...
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
The U.S. Food and Drug Administration (FDA) recently issued a final guidance, “FDA Categorization of Investigational Device Exemption Devices to Assist the Centers for Medicare and Medicaid Services (CMS) with Coverage Decisions”, which modifies the FDA’s policy on categorization of IDE devices used in clinical studies. Devices with an approved Investigational Device Exemption (IDE) are categorized into one of two categories by the FDA — Experimental (Category A) devices or Nonexperimental/Investigational (Category B) devices — based on the level of risk the device poses to the patient. This categorization is used by CMS to determine whether an IDE device, and certain services related to those devices, may be covered by CMS.

When I began practicing as an electrophysiologist in 1994, the dream of successfully using cardiac catheter ablation to ...
West Virginia residents experience disproportionately high instances of cardiovascular disease in comparison to other ...
Change Healthcare Cardiology Hemodynamics is an integrated hemodynamic monitoring system for monitoring vital signs and ...
At the 2017 Radiological Society of North America (RSNA) Annual Meeting, GE Healthcare and NVIDIA announced a series of imaging equipment advances powered by NVIDIA’s artificial intelligence (AI) computing platform. The announcements included the new Revolution Frontier computed tomography (CT) system, advancements to the Vivid E95 4-D Ultrasound and development of GE Healthcare’s Applied Intelligence analytics platform.
Vasorum Ltd, the developer and manufacturer of the novel Celt ACD vascular closure device, has added a 7F-sized Celt ACD device to its range following U.S. Food and Drug Administration (FDA) approval of its premarket approval (PMA) supplement. Celt ACD is indicated for arterial puncture closure in both diagnostic and interventional cardiology and radiology patients. Vasorum said the device offers excellent time to hemostasis in a wide variety of clinical situations. The second-generation Celt ACD devices now available in both the United States and Europe have a new improved delivery system which has been designed to enhance the user experience during deployment.
The U.S. Food and Drug Administration (FDA) announced clearance for NuMed’s Cheatham Platinum (CP) Stent System, which includes the Covered CP Stent and the Covered Mounted CP Stent. The Covered CP Stent is a metal framework covered with cloth to patch a blood vessel tear in patients born with heart defects. The device was approved previously to reopen a blocked or narrowed aorta.


 December 28, 2017
December 28, 2017