October 19, 2018 — Abbott announced today that the HeartMate 3 Left Ventricular Assist Device (LVAD) has received U.S ...

A picture is worth a thousand words, and to patients concerned about their health, detailed images of the coronary ...
Shimadzu Medical Systems USA and Change Healthcare have entered a partnership through which Shimadzu will offer Change Healthcare Cardiology Hemo, along with its Trinias line of angiography systems. The announcement was made at the 2018 Transcatheter Cardiovascular Therapeutics (TCT) annual meeting, Sept. 21-25 in San Diego.
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...

October 17, 2018 — The U.S. Food and Drug Administration (FDA) has reviewed information about potential cybersecurity ...
October 17, 2018 — The U.S. Food and Drug Administration (FDA) has cleared an additional indication for rivaroxaban ...
October 16, 2018 — Abbott announced that its High Sensitive Troponin-I blood test has received CE Mark for distribution ...
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
A new study published in the European Heart Journal shows computed tomography (CT) can be a useful aid in heart team decision-making for complex coronary disease. Results also suggest the state-of-the-art imaging technology may offer a non-invasive diagnostic alternative to conventional coronary angiography. Coronary artery disease (CAD) and its complications are a leading cause of death throughout the world. According to therapeutic guidelines, left main or three vessel diseases represent the most severe, high-risk CAD cases, often requiring coronary bypass graft surgery (CABG) or percutaneous coronary intervention (PCI) as treatment.
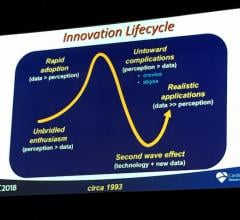
There is a hype cycle surrounding new technologies in all industries, but medicine is unique because of its focus on ...

Sudden cardiac death (SCD) is the leading medical cause of death in young athletes and its impact is consistent worldwide. Most professional athletes in the United States are required to take part in comprehensive cardiovascular screening programs to identify often-asymptomatic congenital or inherited heart disorders, and other cardiac risk factors. There remains a debate however, whether to mandate ECGs as part of pre-participation screening programs for student athletes at the collegiate and high school levels or even at younger ages.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
From a cardiovascular device innovation standpoint, the 2018 Transcatheter Cardiovascular Therapeutics (TCT) conference ...
The nation’s two leading cardiac accreditation and certification organizations are joining forces to offer a single joint product for hospitals beginning Jan. 1, 2019. The American Heart Association and The Joint Commission, the largest standards-setting and accrediting body in healthcare, are collaborating to enhance cardiovascular patient care and improve outcomes and quality of life for patients with cardiovascular disease.
Computed tomography (CT)-based measures of calcification in the abdominal aorta are strong predictors of heart attacks and other adverse cardiovascular events — stronger even than the widely used Framingham risk score. These assertions are according to a new study published in the journal Radiology.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
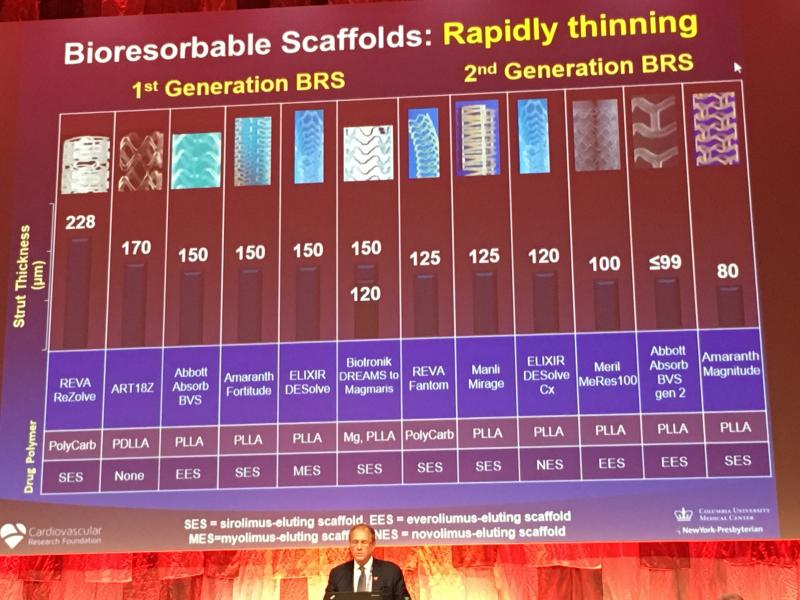
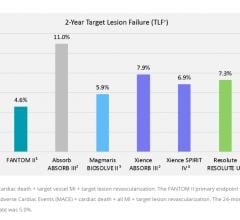
Bioresorbable stent (BRS) technology is not dead, but the unbridled enthusiasm seen two years ago for the technology has ...
October 12, 2018 – Reva Medical presented four key data sets demonstrating the capabilities of the company’s Fantom ...
The Council on Radionuclides and Radiopharmaceuticals Inc. (CORAR) — the voice of the radionuclide, radiopharmaceutical and nuclear pharmacy industries in North America — strongly supports legislation recently introduced by Congressman George Holding (R-NC-2) and Seth Moulton (D-MA-6). This legislation, designated H.R. 6948 - Medicare Diagnostic Radiopharmaceutical Payment Equity Act of 2018, would direct the Secretary of Health and Human Services to recognize diagnostic radiopharmaceuticals (RPs) as drugs and institute a separate payment policy under the Medicare Hospital Outpatient Prospective Payment System (OPPS).

 October 19, 2018
October 19, 2018