May 16, 2022 — Right heart failure (RHF) continues to be a major source of morbidity and mortality following left ...
May 16, 2022 — The Cardiovascular Research Foundation (CRF) announced that TVT: The Structural Heart Summit will feature ...
May 13, 2022 — Bristol Myers Squibb’s mavacamten will stand rather unchallenged in the hypertrophic cardiomyopathy (HCM) ...
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
May 13, 2022 — Acutus Medical, an arrhythmia management company focused on improving the way cardiac arrhythmias are ...
May 13, 2022 — The MemorialCare Heart & Vascular Institute at Long Beach Medical Center has been designated the only ...
May 13, 2022 — Vascular diseases are public enemy number one: the leading killers worldwide, accounting for nearly a ...
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
May 12, 2022 — Abbott announced a new partnership with Women as One to launch a new program designed to train more ...
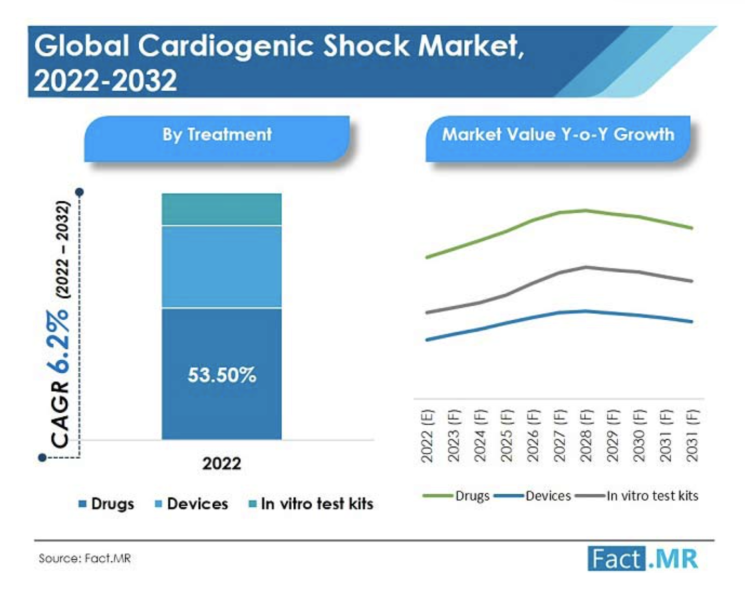
The global cardiogenic shock market was valued at US$ 3.29 Bn in 2021, and is expected to reach US$ 6.32 Bn by 2032 ...
May 12, 2022 — Vivalink, a leading provider of digital healthcare solutions, announced availability of the world’s first ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
May 12, 2022 — NDA Partners General Manager, Eric Fish, announced today that Peter Mackrell, MD, a board certified ...
May 11, 2022 — Women who experienced complications related to developing high blood pressure, or hypertension, during ...
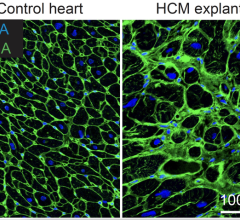
May 11, 2022 — Hypertrophic cardiomyopathy is a heart disease that leads to a stressed, swollen heart muscle. Due to a ...
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
May 11, 2022 — Vital Heart & Vein, a leading provider of cardiovascular and vein health, has named Gay Nord as its new ...

In April, the cardiology community converged face-to-face in Orlando for The American College of Cardiology’s (ACC22) ...
May 10, 2022 — The MIVI Neuroscience Q Aspiration Catheter incorporates a novel pusher wire design on its proximal end ...


 May 16, 2022
May 16, 2022