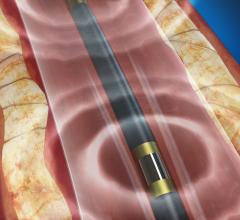
Claret Medical announced its filing of a marketing application with the U.S. Food and Drug Administration (FDA) for clearance of the Sentinel Cerebral Protection System (CPS). The Sentinel CPS is designed to protect the brain by capturing and removing debris dislodged during transcatheter aortic valve replacement (TAVR) that enters cerebral circulation and carries the potential for stroke. There are currently no cerebral embolic protection technologies available in the United States to protect TAVR patients from cerebral embolic events, according to Claret Medical.
Medtronic plc announced the results of an analysis that reveals patients increasingly adhere to heart failure medications after they receive cardiac resynchronization therapy (CRT) devices. The analysis of more than 4,500 patients revealed that compliance with their medications increased 67 percent 24 months after receiving their CRT implants, compared to the 24 months prior to implant (p<0.001). The results were presented at the 2016 Heart Failure Society of America (HFSA) Scientific Meeting in Orlando, Fla.
September 20, 2016 — Teleflex Inc. recently displayed its vascular access technologies and a new educational platform ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
September 19, 2016 — Edwards Lifesciences received European CE mark to expand use of the Edwards Sapien 3 transcatheter ...
September 19, 2016 — Shockwave Medical announced positive clinical results from the pooled DISRUPT PAD Study, a single ...
Valtech Cardio Ltd. announced that it will present the first-in-man data for its Cardioband Tricuspid (TR) system at PCR London Valves 2016, Sept. 18-20. In addition, updated two-year follow-up results of the multi-center Cardioband Mitral study will be presented at the meeting.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
September 19, 2016 — Boston Scientific received European CE mark approval for its Lotus Edge Valve System, the company's ...
Medtronic plc announced that the U.S. Food and Drug Administration (FDA) approved the IN.PACT Admiral drug-coated balloon (DCB) as a treatment for in-stent restenosis (ISR) in patients with peripheral artery disease (PAD). This is the first DCB that has gained approval to treat ISR in the United States. FDA approval was based on ISR data from the IN.PACT Global Study compared to a standard percutaneous balloon angioplasty (PTA) control.
September 16, 2016 — The U.S. Food and Drug Administration (FDA) has granted market clearance for Shockwave Medical’s ...
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
This video, provided by Shockwave Medical, demonstrates the Lithoplasty System. It uses ultrasonic waves to treatment ...
EBR Systems Inc. announced the U.S. Food and Drug Administration (FDA) has granted an Investigational Device Exemption (IDE) for its WiSE (Wireless Stimulation Endocardially) Technology for cardiac resynchronization therapy (CRT). This IDE enables EBR Systems to start a major U.S. study to establish safety and effectiveness in support of U.S. approval.

Vascular Interventional Advances (VIVA) 2016 released its list late-breaking research presentations in vascular ...
Change Healthcare Cardiology Hemodynamics is an integrated hemodynamic monitoring system for monitoring vital signs and ...
September 13, 2016 — Results from a new study suggest that small molecules known as microRNAs may be part of the pathway ...
September 13, 2016 — Findings from a small study suggest that people who followed the popular Paleo diet for only eight ...
(Editor’s note: This is the first part of a two-part series on the proposed Medicare five-year demonstration for ...


 September 20, 2016
September 20, 2016