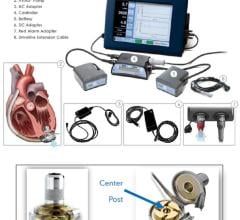
CircuLite Inc. reported the launch of the clinical development program for its Synergy Pocket Circulatory Assist Device with the successful implantation of the first patient in a European feasibility trial. Synergy is a miniature implantable blood pump, the size of a AA battery, that can be implanted superficially in a pocket, like a pacemaker.
The device is designed to provide long-term, partial circulatory support in patients with chronic heart failure. The primary objective of the first-in-man trial is to assess the safety of the device in patients with chronic heart failure who are waiting to receive heart transplants. Bart Meyns, M.D., Ph.D., professor and chief of Cardiac Surgery at Gasthuisberg University Hospital in Leuven, Belgium, performed the first implant. CircuLite’s ultimate goal is to expand the treatment of heart failure to the chronic, ambulatory patient in order to improve their quality of life by giving them an elective, less-invasive option to increase blood flow from the heart.


 June 19, 2024
June 19, 2024