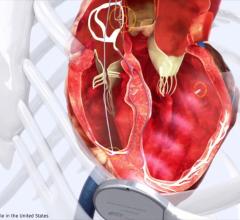
May 9, 2017 — The U.S. Food and Drug Administration (FDA) has cleared Boston Scientific’s Resonate family of implantable cardioverter defibrillator (ICD) and cardiac resynchronization therapy defibrillator (CRT-D) systems. The approval includes new features in the Resonate devices including SmartCRT technology with multisite pacing capability for multi-electrode pacing, and compatibility with the HeartLogic Heart Failure Diagnostic Service to help physicians improve heart failure (HF) management.
All Resonate devices are powered by the company's EnduraLife battery technology, which has nearly two times the usable battery capacity as certain competitive devices,[1,2] an essential factor when considering the lifelong needs of patients receiving ICD or CRT therapy. Recent guidance issued by the National Institute for Health and Care Excellence (NICE) in March 2017 recommended the use of EnduraLife-powered CRT-Ds to reduce the number of avoidable replacement procedures a patient may have to undergo, thereby offering improved outcomes for patients and potential savings to the National Health Service (NHS) in England of approximately £6 million in the first five years.
The company has initiated a series of clinical trials, the SMART Registry, SMART CRT Study and SMART MSP, to demonstrate improved response to CRT therapy with SmartCRT technology, which helps physicians optimize where, when, and how to pace the lower chambers of the heart.
"These trials will add to the body of evidence showing CRT therapy can be tailored to individual patient characteristics at the time of implant, while adjusting device programming solutions over the life of the device without fear of adversely draining the device battery and causing unnecessary replacement procedures," said Michael Gold, M.D., principal investigator of the SMART CRT study and the Michael E. Assey professor of medicine at The Medical University of South Carolina, Charleston, S.C.
"We are changing the treatment landscape by combining industry-leading device longevity with innovative solutions that will provide clinicians with tools to manage heart failure more effectively," said Kenneth Stein, M.D., senior vice president and chief medical officer, rhythm management and global health policy, Boston Scientific. "Our post-approval studies for the HeartLogic service, including MANAGE-HF, will gather additional evidence to illustrate how these alerts, which detect impending heart failure decompensation, can improve patient outcomes."
This FDA approval follows on the February 2017 CE mark and subsequent commercialization for the Resonate family of ICD and CRT-D devices.
For more information: www.bostonscientific.com
References:
1. Medtronic Evera XT VR DVBB2D4 Device Manual page 25.
2. Boston Scientific Implantable Cardioverter Defibrillator Physician's Technical Manual 359050-003 EN US 2014-01 page 30.


 July 21, 2025
July 21, 2025