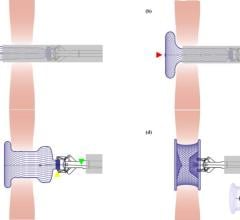
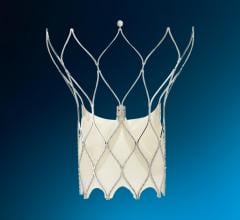
The Watchman device is designed to keep harmful sized blood clots that form in the left atrial appendage from entering the blood stream, potentially causing a stroke. The device is permanently placed just behind or at the opening of the left atrial appendage.
Using standard techniques, similar to the ones used in commonly performed angioplasty procedures, the Watchman device is guided into the heart through a catheter. Once the catheter is in the correct position, pictures of the heart are taken in order to take appropriate measurements of the left atrial appendage. The measurements will determine which size Watchman device to use.


 June 20, 2024
June 20, 2024