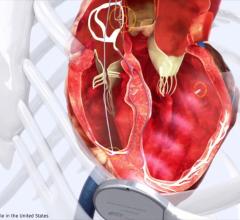
March 23, 2017 — Biotronik announced the European launch of the Edora series, its smallest series of pacemakers and cardiac resynchronization therapy pacemakers (CRT-Ps). The new devices are equipped with features geared towards improving patient safety, therapy and comfort. Moreover, Edora pacemakers are significantly smaller and lighter than preceding models, achieving compactness and full functionality without compromising battery life.
“Biotronik’s new series of pacemakers fits well in the current age of smart technologies, with devices that are smaller, lighter and shorter while retaining longevity,” stated Saravanan AL Krishinan, M.D., Hospital Sultanah Bahiyah, Alor Setar, Malaysia. "What's more, the devices offer simplified MRI [magnetic resonance imaging] access, which is often crucial for better patient care.”
To facilitate patient access to MRI, Edora pacemakers and CRT-Ps are MR-conditional and feature Biotronik’s MRI AutoDetect functionality. This means that within a programmable window of up to 14 days, the device’s built-in sensor can detect an MRI environment and automatically switch the system to MRI mode; the device then automatically switch back to its standard mode once the scan is complete. Since the device is only in MRI mode for the duration of the scan, the patient receives optimal therapy for the maximum amount of time.
Devices in the Edora series also support Biotronik Home Monitoring for remote long-term patient care with a streamlined workflow. Biotronik Home Monitoring automatically collects cardiovascular data from the patient’s device on a daily basis and transmits it remotely to the physician without the need for any patient interaction. This facilitates the early detection of arrhythmias, with clinical trial results demonstrating that the monitoring system significantly reduces hospitalization for atrial arrhythmia and related stroke in pacemaker patients.
Edora CRT-Ps are specifically tailored for individualized therapy through quadripolar technology for flexible pacing in the left ventricle (LV). This is complemented by a comprehensive LV lead portfolio, which caters for diverse patient anatomies.
This product is currently only approved for use in the European Economic Area. The company expects to announce FDA approval in the second quarter of 2017.
For more information: www.biotronik.com


 July 21, 2025
July 21, 2025