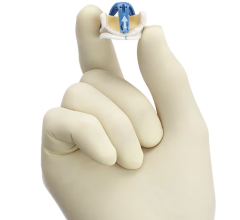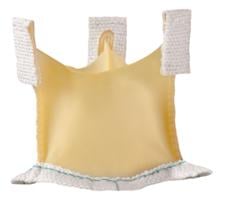
June 11, 2014 — Medtronic Inc. announced five-year follow-up data demonstrating the safety and performance of the Medtronic 3f Enable Aortic Bioprosthesis — the world's first commercially available sutureless tissue heart valve. The longest follow-up data ever reported for a sutureless bioprosthesis, results showed that patients implanted with 3f Enable displayed no structural valve deterioration at five years, and demonstrated 93 percent freedom from valve mortality (death related to valve complications) at five years post-implant, a performance comparable to traditional surgical heart valves. The results were presented at the 94th American Association for Thoracic Surgery Annual Meeting in Toronto. Medtronic 3f Enable received CE mark in December 2009. It is not approved for use in the United States, Canada or Japan.
The Medtronic 3f Enable Bioprosthesis facilitates implantation using a less-invasive surgical technique to replace diseased, damaged or malfunctioning native or prosthetic aortic valves in patients with aortic valve stenosis or aortic insufficiency, conditions that affect more than 11.6 million people worldwide.[1] Implantation is possible through an incision that is smaller than those commonly used for traditional surgical valve replacements, and without conventional sutures, which potentially reduces procedure time and makes it easier on the patient.[2],[3] The valve's self-expanding Nitinol frame allows the 3f Enable valve to be folded into a small diameter size so that it fits through the minimal incision. The valve is fixated in the valve's annulus (opening) and can be repositioned as needed.
In the study, patients five years post-implant also had consistently low (single-digit) gradients — a measure of blood flow indicating that blood is able to flow through the valve without interference.
"Until now, we didn't know the medium- to long-term durability for this new device for fast aortic valve replacement," said Prof. Thierry Carrel, M.D., director of the department of cardiovascular surgery at the University Hospital Berne in Switzerland. "It's very important that this sutureless valve performs very well, long after it's implanted. Patients and physicians alike should be encouraged by this data."
"This is the first time we've seen five-year performance data of any sutureless valve on the market, and the results are quite impressive," said John Liddicoat, M.D., senior vice president, Medtronic, and president of the Structural Heart Business. "The 3f Enable tissue heart valve not only provides a less invasive way to replace diseased, damaged or malfunctioning aortic valves, but now we know it performs well over time with excellent gradients, too."
For more information: www.medtronic.com
References:
1. 2005 Health Research International Report
2. Eichstaedt et al, Early single-center experience in sutureless aortic valve implantation in 120 patients (J Thorac Cardiovasc Surg 2013;-:1-6)
3. Martens et al, Eur J Cardiothorac Surg 2011;40:749-55


 January 15, 2026
January 15, 2026