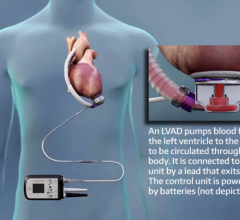
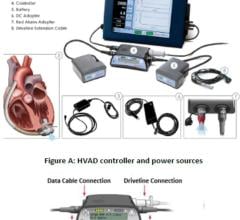
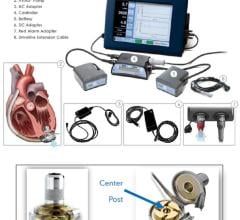
March 18, 2014 — Thoratec Corp. initiated a voluntary worldwide Medical Device Correction in order to update its labeling and training materials for the HeartMate II LVAS Pocket System Controller. The following information is provided as a reinforcement of the initial release.
Failure to completely connect the driveline during the process of exchanging Pocket System Controllers has resulted in serious injuries and deaths. Patients who received the Pocket Controller as a replacement for an older model (EPC System Controller) may be at a higher risk of experiencing difficulty in the controller exchange process, as there are differences between the two controller models, especially differences related to the connection of the driveline. As of Feb. 4, 2014, the rate of serious injury or death during the process of controller exchange for patients who converted to the Pocket Controller from the EPC Controller was 1.5 percent (8/523), while the rate for patients who received the Pocket Controller at the time of implant was 0.1 percent (1/1619).
Patients who are using the HeartMate II LVAS Pocket Controller should immediately contact their doctor for retraining on use of the device and to receive updated Patient Handbook information if they have not been contacted by their doctor already. Pocket Controllers can be identified by the following model information that shows on the label on the controller: REF 105109.
Adverse reactions or quality problems experienced with the use of this product may be reported to the U.S. Food and Drug Administration (FDA)'s MedWatch Adverse Event Reporting program either online, by regular mail or by fax.
- Online: www.fda.gov/medwatch/report.htm
- Regular Mail: use postage-paid FDA form 3500 available at: www.fda.gov/MedWatch/getforms.htm. Mail to MedWatch 5600 Fishers Lane, Rockville, MD 20852-9787
- Fax: 1-800-FDA-0178
For more information: www.thoratec.com


 June 19, 2024
June 19, 2024