April 17, 2012 – The U.S. Food and Drug Administration (FDA) has approved a humanitarian use device (HUD) designation for the SynCardia temporary Total Artificial Heart to be used for destination therapy in addition to its current PMA approval as a bridge to transplant.
“This is a huge milestone that with approval of our HDE will allow the Total Artificial Heart to save the lives of up to 4,000 U.S. patients each year who are not eligible for transplant,” said Michael Garippa, SynCardia chairman/CEO/president.
The FDA approval letter of the HUD request designates the Total Artificial Heart for use in U.S. patients “at risk of imminent death from non-reversible biventricular heart failure who are not eligible for cardiac transplant and have a body surface area (BSA) of ? 1.7m2.”
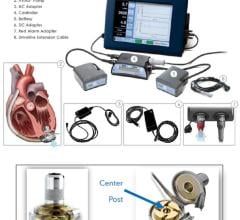
“Historically, the Total Artificial Heart had been limited to temporary use because U.S. patients couldn’t leave the hospital with the 400-pound driver,” said Garippa. “However, the new 13.5-pound Freedom portable driver* has leveled the playing field in mechanical circulatory support. By making patient discharge possible, the Freedom driver has made the Total Artificial Heart a viable option to support patients for the rest of their lives while allowing hospitals to make the best choice for each individual patient based on their medical needs.”
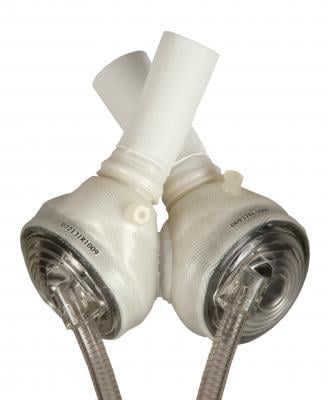
Originally used as a permanent replacement heart, the SynCardia Total Artificial Heart received FDA approval in 2004 as a bridge to transplant for transplant-eligible patients dying from end-stage biventricular heart failure. Currently, the longest a patient has been supported with the Total Artificial Heart is 1,374 days (almost four years) prior to receiving his heart transplant.
During 30 years of use, the valves in the SynCardia Total Artificial Heart have never failed. The diaphragm has a failure rate of less than 1 percent over more than 1,000 implants (2,000-plus diaphragms).
*The Freedom portable driver is the world’s first wearable power supply for SynCardia’s Total Artificial Heart. It is CE approved for use in Europe and undergoing an FDA-approved investigational device exemption (IDE) clinical study in the United States.
For more information: www.syncardia.com


 June 19, 2024
June 19, 2024