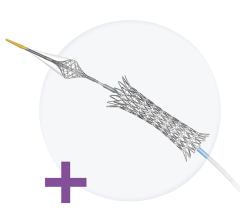
June 4, 2007 — Biodegradable magnesium stents have been developed to treat blockages in coronary arteries. The stents will degrade within four months and achieve the same results as conventional stents, conclude authors of an article published in this week's edition of The Lancet.
But both the article and an accompanying comment warn longer term follow-up of the patients fitted with biodegradable stents is needed.
Coronary stents improve immediate and late results of balloon angioplasty by closing up dissections and preventing collapse of the arterial walls.
Professor Raimund Erbel, Department of Cardiology, West German Heart Centre Essen, Germany, and colleagues successfully implanted 71 of the biodegradable magnesium stents in 63 patients in their study the PROGRESS-AMS clinical trial.
The researchers found that after 12 months, the stents were safe, with no incidence of stent thrombosis (clotting within the stent), heart attack or death in any of the patients. They found that the diameter of the blood vessel within the stent (in-stent acute gain) increased by an average of 1.41mm. Whilst the stent struts disappear over time, ultrasound confirms they remain present but have been absorbed into the vessel walls, with the space left behind "filled in." The magnesium is eventually replaced by calcium and phosphorous through the body's natural processes.
However the trial also showed that angiographic restenosis (re-blocking of the artery) occurred in 47.5% of patients who received the biodegradable stents, and 27% needed target lesion vascularisation within 12 months due to recurrent ischaemia (lack of blood flow to the tissues).
The authors said, "This study showed that absorbable magnesium stents can be delivered and expanded at high pressure in artherosclerotic coronary arteries, providing good mechanical scaffolding and achieving a lumen enlargement similar to the immediate lumen gain obtained with conventional metallic stents."
The article explains that the new biodegradable stents overcome the limitations of conventional permanent stents namely that they can cause thrombosis many months after implantation and thus regular dual antiplatelet drug treatment such as aspirin and clopidogrel are required to prevent this occurring in patients with conventional stents fitted.
Drug-eluting stents have limitations because despite being able to achieve significant decreases in angiographic restenosis, they can cause hypersensitivity reactions and late-stent thrombosis, possibly because the drugs eluted hinder re-endothelialisation (re-lining of the inside wall of the artery) of stent struts. Biodegradable stents avoid this problem.
The authors caution that studies with more patients and longer term follow-up are required to confirm the safety of biodegradable magnesium stents.
"For a new product such as absorbable metal stents, four months of angiographical study and 12 months clinical follow-up might be insufficient to capture all the potential late events and complications."
"This study shows that biodegradable magnesium stents can achieve an immediate angiographic result similar to the result of other metal stents and can be safely degraded after four months,” conclude the authors. “Modifications of stent characteristics with prolonged degradation and drug elution are currently in development."
In an accompanying comment, Dr John Ormiston and Dr Mark Webster, Mercy Angiography and Auckland City Hospital, Auckland, New Zealand, said, "Bioabsorbable stents are in an early stage of development but hold considerable promise for overcoming many of the limitations of permanent metallic implants."
They add, "The initial patients treated with these devices will need to be followed up closely and for a long time. Whether bioabsorbable stents signal the threshold of a new era for percutaneous coronary intervention is yet to unfold."
For more information visit: www.thelancet.com


 November 24, 2025
November 24, 2025