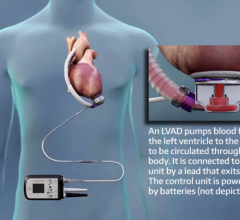
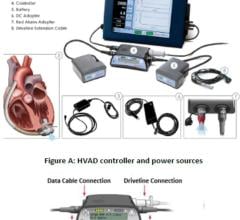
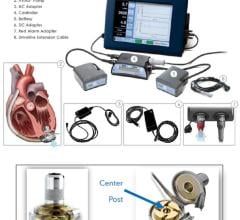
May 1, 2007 — Ventracor has announced the presentation of summary clinical data of the first 100 patients implanted with the VentrAssist third generation left ventricular assist device (LVAD), an implantable blood pump designed for long term use in patients with advanced heart failure.
To date, 108 patients have been implanted with the VentrAssist, for over 50 patient years of support, and 42 patients are currently supported — the longest duration is two years and eight months. This is greater experience than all other third generation centrifugal LVADs combined, the company says. The VentrAssist is market released in Europe (CE Mark) and is the subject of two U.S. clinical trials for Bridge to Transplant (BTT) and Destination Therapy (DT) indications.
Summary clinical data from the first 100 VentrAssist patients was presented Monday at a plenary session of the 2007 annual meeting of the International Society of Heart and Lung Transplantation (ISHLT) by Professor Don Esmore of the Alfred Hospital, in Melbourne, Australia.
As a pioneer in the field of LVADs, Professor Esmore has the most clinical experience with and was the first surgeon in the world to implant the VentrAssist.
A full copy of the presentation is available at www.ventracor.com.
"The VentrAssist has changed the lives of many people, and expanded the choices for treatment of end stage heart failure. Full results of the 33 patients enrolled in the CE Mark Bridge to Transplant trial showed a survival rate of around 85 percent," Professor Esmore told the international audience of heart failure specialists.
"Since the first implant in 2003, we have learned much about patient selection and support, and we expect that future clinical trials will continue to show improvement in patient outcomes," Professor Esmore added.


 June 19, 2024
June 19, 2024