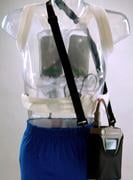
October 6, 2010 – Physicians have prescribed more than 30,000 wearable defibrillator vests for patients at high risk of sudden cardiac arrest (SCA). The LifeVest Wearable Defibrillator, by Zoll Medical, gives a physician time to assess the patient’s long-term arrhythmic risk and make appropriate plans.
The LifeVest is lightweight and easy to wear and allows patients to return to their normal daily living. It continuously monitors the patient’s heart, and if a life-threatening heart rhythm is detected, the device delivers a treatment shock to restore the normal rhythm.
The device can be used following a heart attack, before or after bypass surgery or stent placement, and in those with cardiomyopathy or congestive heart failure.
Most health plans in the United States cover the LifeVest.
For more information: www.zoll.com


 January 13, 2026
January 13, 2026 









