Thoratec's HeartMate II
February 16, 2009 - Thoratec Corp. and HeartWare International last week entered into a definitive merger agreement under which Thoratec will acquire HeartWare for a about $282 million, and the combined company will offer a broad portfolio of approved blood pumping devices for the treatment of heart failure.
The companies said approximately 50 percent of the purchase price will be paid in cash and about 50 percent will be paid in shares of Thoratec common stock. Based on a Thoratec common stock price of $26.25 per share, this reflects a current price of $0.86 for each HeartWare Chess Depositary Interest (CDI), or $1.32-Austrialian based on the current U.S./AUS exchange rate of 1.5265.
"This transaction is a positive development for heart failure patients and the clinicians who treat them by combining Thoratec’s portfolio of commercially approved devices with HeartWare’s innovative technologies,” said Gary F. Burbach, president and chief executive officer of Thoratec. “The use of mechanical circulatory support for the treatment of heart failure is gaining increasing adoption as a result of the positive patient outcomes and clinician enthusiasm realized with the HeartMate II. We believe that combining the strengths of the two companies will enable us to build upon each of our strong technology and product platforms, giving more and better options for a large and significantly underserved heart failure patient population.
HeartWare recently received the CE Mark for the HeartWare Ventricular Assist System and said it is experiencing strong initial progress in its U.S. BTT trial. The company is also working on its MVAD, a next generation miniaturized axial flow LVAD (left ventricular assist device).
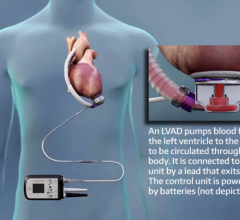
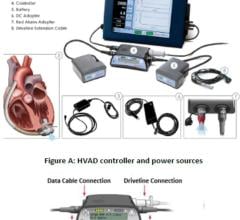
Thoratec’s product line includes several commercially approved cardiac assist devices including the HeartMate II LVAS, which received U.S. approval for bridge-to-transplantation (BTT) in April 2008 and is currently in clinical trials in the U.S. for destination therapy (DT) or the long-term support of heart failure patients not eligible for transplantation.
Upon the close of the transaction, HeartWare's operations will be integrated into Thoratec's Cardiovascular Division. Further details regarding the nature and timing of the integration will be provided in the future.
For more information: www.thoratec.com, www.itcmed.com


 June 19, 2024
June 19, 2024