July 13, 2011 – The TandemHeart circulatory support system has been granted expanded reimbursement coverage by NHIC Corp., the medicare administrative contractor (MAC) serving over 1.3 million beneficiaries and over 53,000 healthcare providers in the Northeast region of the United States.
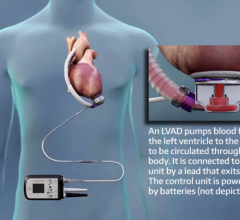
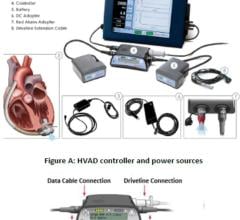
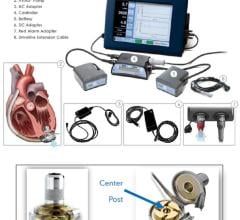
Physicians have utilized the TandemHeart device to provide critical heart support to over 2,500 patients at more than 170 of the top cardiac centers in the United States. The system is composed of a small centrifugal pump, a series of cannulae that connect the pump to the patient and an electro-mechanical controller. Starting from a small incision in a patient's leg, cannulae are inserted into the blood vessels and up to the heart. The TandemHeart pump is then activated, allowing damaged heart muscle to rest while augmenting the flow of blood to the patient's vital organs and the rest of the body.
NHIC's local coverage determination (LCD) for percutaneous ventricular assist devices (VAD) became effective July 8, 2011, and provides coverage of TandemHeart as a primary treatment option for a wide range of indications. It applies to Medicare beneficiaries throughout NHIC's jurisdiction, including those located in Maine, Massachusetts, New Hampshire, Rhode Island and Vermont.
The U.S. Food and Drug Administration (FDA)-cleared TandemHeart system is the only heart assist device that can provide a high degree of circulatory support with a minimally invasive, percutaneous procedure. With flow rates up to five liters per minute, the TandemHeart delivers as much as five times the hemodynamic support as the standard of care intra-aortic balloon pump and twice the support of any other available percutaneous technology.
For more information: www.cardiacassist.com


 June 19, 2024
June 19, 2024