October 6, 2014 — St. Vincent Heart Center officials announced that the faith-based hospital is the second in the nation to implant the new investigational “heart pump.” Christopher Salerno, cardiothoracic surgeon at St.Vincent Heart Center implanted the new Left Ventricular Assist System (LVAS) called the HeartMate III on a 19-year-old patient. St. Vincent Heart Center is participating in the U.S. Investigational Device Exemption (IDE) clinical trial with the HeartMate III for patients with congestive heart failure. The Thoratec HeartMate III Left Ventricular Assist Device (LVAD) is a new advancement in mechanical circulatory support (MCS). St. Vincent Heart Center is part of St. Louis-based Ascension Health.
Lucas Jackson, 19, of North Vernon, Ind., was diagnosed with hypertrophic cardiomyopathy — a disease in which the heart muscle becomes abnormally thick, making it harder for the heart to pump blood — and congestive heart failure days after his 18th birthday. Nine months earlier, his father passed away from the same condition. Prior to his father’s passing and his diagnosis, Jackson was your typical teenager with a passion for dirt track car racing. The oldest of four, he is one of three siblings who have a genetic heart condition. Jackson received the new HeartMate III LVAD while he waits for a new heart.
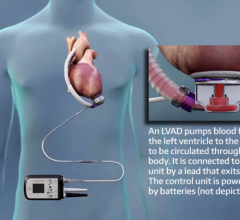
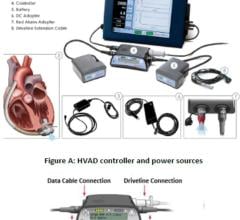
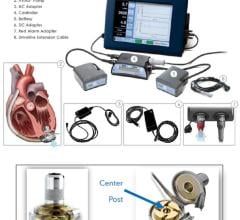
The HeartMate III LVAD is a small implantable mechanical device that helps circulate blood throughout the body. Sometimes called a “heart pump” or “VAD,” it is designed to supplement the heart’s pumping function for patients whose hearts are too weak to adequately pump blood on their own. The fully magnetically levitated technology foundation is designed to lower adverse event rates through improved compatibility with blood, while HeartMate III also enhances ease of surgical placement through its compact size.
Compared to other LVADs on the market, the HeartMate III has been designed to be more patient friendly. The battery has a longer run time — thus far testing shows 17 hours on a pair of batteries. Also, the smaller device allows for a less invasive surgery. Some of the benefits of this new device are that there is no pump pocket required below the diaphragm thus potential for less dissection, shorter surgery time, and less bleeding.
“While the number of patients being diagnosed with advance heart failure continues to rise, the number of donor hearts has remained stagnant,” said Salerno. “St. Vincent Heart Center is proud to be one of the first to assess this therapy and look forward to making it available to patients in Indiana.”
More than 5 million people in the United States are afflicted with congestive heart failure (CHF). Of these, more than 250,000 are advanced cases, with an annual mortality rate of 80 percent.
The HeartMate III LVAS U.S. IDE Clinical Trial is designed to evaluate the safety and effectiveness of the investigational device. Patients who agree to participate in the study are randomly assigned to receive either the HeartMate II or the HeartMate III LVAD.
For more information: 1.usa.gov/YcZfqb


 June 19, 2024
June 19, 2024