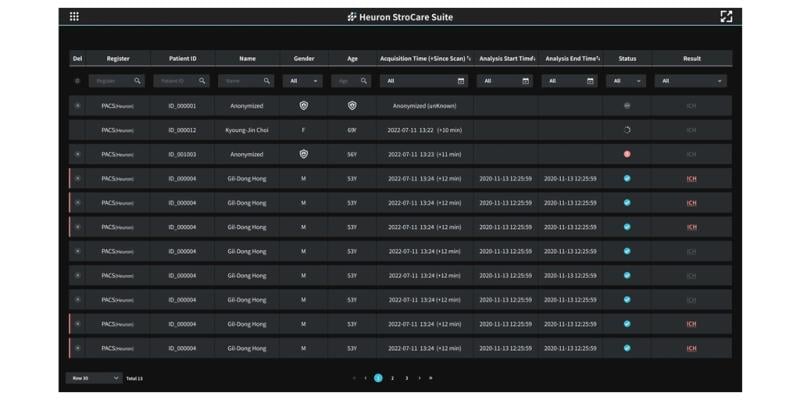
Heuron ICH Worklist (Graphic: Business Wire)
June 3, 2024 — Heuron, a specialized medical AI startup focused on brain and neurological disorders based in South Korea, announced that its non-contrast CT imaging analysis solution, Heuron ICH, has received FDA 510(k) clearance. This marks the first FDA approval of an AI-based brain disorder analysis solution from Korea for computer-aided triage and notification devices (CADt).
Heuron ICH is an advanced AI solution designed to automatically screen and signal potential cases of intracranial hemorrhage, relying solely on non-contrast CT images. Its primary function is to swiftly discern emergency brain hemorrhages that require immediate attention, even capturing the minutest hemorrhages that often elude human detection. Notably, Heuron ICH boasts a sensitivity of 86% and specificity of 88%, ensuring reliable and accurate assistance in critical medical scenarios.
Heuron ICH becomes the fifth solution from Heuron to receive FDA clearance, following Veuron-Brain-mN1, Veuron-Brain-pAb, Heuron Beta Amyloid, and Veuron-Brain-pAb3. Currently, Heuron holds 15 domestic medical device approvals/certifications, four European CE (MDD) certifications, and various other international certifications.
Donghoon Shin, CEO of Heuron, expressed, "Securing this product approval marks a significant milestone, facilitating our swift penetration into the U.S. market. Our commitment extends beyond this achievement as we endeavor to expand our portfolio of FDA-approved medical solutions. We are dedicated to optimizing the utilization of Heuron's diverse offerings within U.S. clinical environments, particularly for emergency patient care."
In addition, Heuron ICH successfully achieved the performance levels required by the FDA through clinical trials conducted last year in collaboration with Mass General Brigham, affiliated with Harvard Medical School.
For more information: www.iheuron.com/en/


 February 02, 2026
February 02, 2026