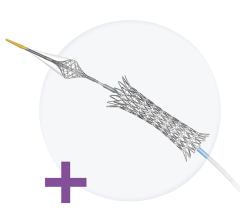
February 22, 2010 – During the Joint Interventional Meeting (JIM) Feb. 11-13 in Rome, the Cappella Sideguard 3.25 bifucation stent was highlighted.
Gary S. Mintz, M.D., Cardiovascular Research Foundation, New York, presented “Positive Vessel Remodeling Using Sideguard in Coronary Bifurcations” during the new technologies symposium. Dr. Mintz highlighted that serial IVUS and OCT studies of the stent are ongoing in the Sideguard 3 clinical trial to confirm the initial findings of Sideguard 1 and assess stent strut coverage. The IVUS substudy results from Sideguard 1 were published in the October 2009 issue of the American Journal of Cardiology.
The newest addition to the Sideguard portfolio in Europe, the Sideguard 3.25, was implanted successfully during a live case performed by Ralf Mueller, M.D., Siegburg, Germany. “We implanted the Sideguard 3.25 stent together with a 3 mm DES in a medina 0,1,1 LAD/D1 stenosis,” he said. “Positioning was easy and precise and IVUS demonstrated excellent scaffolding at the bifurcation. The use of the Sideguard device made this bifurcation intervention easy and predictable with an optimal acute result.”
Martin B. Leon, M.D., founder and director of Transcatheter Cardiovascular Therapeutics (TCT) and Scientific Advisory Board member of Cappella assisted with the case. Dr. Leon expressed his satisfaction with the device, “It was the first time I used the 3.25 and I was very pleased with the outcome of the case.”
For more information: www.cappella-med.co


 November 24, 2025
November 24, 2025