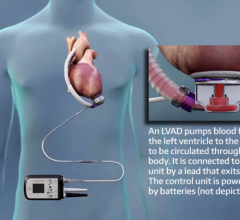
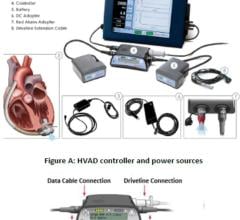
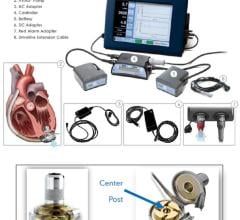
January 7, 2008 - Abiomed Inc. has rolled out a new Portable Circulatory Support Driver for both in-hospital and out-of-hospital patients designed to support Abiomed’s AB5000 Ventricular Assist Device (VAD), designed to provide either univentricular or biventricular support.
The combination of Abiomed’s new Portable Driver and its FDA-approved AB5000 VAD is engineered to provide support of acute heart failure patients. In many cases, profound shock patients require biventricular support (both sides of the heart). The AB5000 can assume the pumping function of a patient’s failing heart, allowing the heart to rest, heal and potentially recover. AB5000 is designed to provide either univentricular or biventricular support. Abiomed also announced today that it has received FDA labeling approval of one-year bench reliability for its AB5000 VAD, which is expected to complement the Portable Driver reliability.
“My experience with the AB5000 Circulatory Support System has been excellent in treating over 30 patients with support duration of up to 40 days,” said Louis Samuels, M.D., FACS Surgical Director Heart Failure and Transplant, director Artificial Heart & Ventricular Assist Device Program, Lankenau Hospital. “The new Portable Driver is lightweight and quiet, demonstrates reliable performance, and has the potential to improve patient care, while lowering costs to hospitals.”
Abiomed’s Portable Driver is a biventricular system, which delivers the pressures and vacuums equivalent to Abiomed’s AB5000 console and its recently approved iPulseT console, at only 18 pounds in weight. The unit is expected to require low maintenance, approximately every 5,000 hours of operation, which is estimated to be three times longer than existing portable consoles that weigh twice as much. Abiomed’s Portable Driver was designed with the latest smart battery technology for extended power capability and the quiet operation of the Portable Driver provides for minimal disruption of the patient’s quality of life at-home or while in care centers.
“There is a clinical need to provide patients requiring VAD support a greater degree of mobility during treatment aimed at myocardial recovery,” said John V. Conte, M.D., associate director of Cardiac Surgery, associate professor of Surgery, director of the Heart and Lung Transplantation, director of the Ventricular Assist Device Program, Johns Hopkins Hospital. “The expected durability of this portable driver is a nice match with the AB5000 VAD and could provide an integrated and effective solution for patients requiring univentricular or biventricular support.”
Abiomed’s Portable Driver has not yet been approved by the U.S. FDA. The Company plans to pursue an investigational device exemption (IDE) to conduct a discharge study, which is expected to be submitted in early calendar 2008. The AB5000 VAD is approved in Europe under CE-mark.
For more information: www.abiomed.com


 June 19, 2024
June 19, 2024