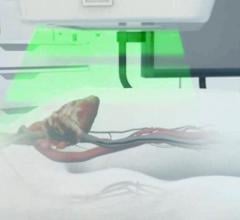
October 29, 2012 — Boston Scientific Corporation has received regulatory approval to market the Reliance 4-Front lead ...
EP Lab
This channel includes news and new technology innovations for cardiac electrophysiology (EP) systems, techniques and devices using in EP labs. This includes implantable EP devices, pacemakers, implantable cardioverter defibrillators (ICD), cardiac resychronization therapy (CRT), ablation technologies, left atrial appendage (LAA) occlusion, atrial fibrilation (AF) and Holter monitors.
October 26, 2012 — Biotronik began its European market release of BioMonitor, an implantable cardiac device designed for ...
October 19, 2012 — Boston Scientific Corp. entered into a definitive agreement to acquire privately-held Rhythmia ...
When the patients of Michael Boler, M.D. need cardiac monitoring, the Holter monitor is no longer his first choice. “The ...
October 15, 2012 — Cardiac Science has unveiled its Burdick 4250 Holter recorder — a wearable heart monitor half the ...
October 12, 2012 — Biotronik last month announced the start of the prospective multicenter PARCADIA study. The study ...
October 12, 2012 — Estech, a provider of minimally invasive cardiac ablation devices, last month announced the market ...
Apple created a stir when it announced in 2018 that its Apple Watch Series 4 was the first consumer health and fitness ...
October 12, 2012 — TyRx Inc. said it received a license from Health Canada to market its AigisRx Antibacterial Envelope ...
October 8, 2012 — The U.S. Food and Drug Administration (FDA) has granted Boston Scientific Corp. regulatory approval ...
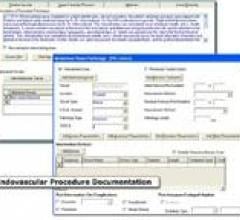
October 8, 2012 — St. Jude Medical Inc. has launched MediGuide Technology, the first 3-D navigation system intended for ...
Washington Health System (WHS) provides healthcare services at more than 40 offsite locations across three counties in ...
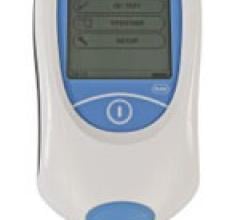
October 8, 2012 — The U.S. Food and Drug Administration (FDA) has granted Clinical Laboratory Improvement Amendments ...
October 3, 2012 — Royal Philips Electronics announced it is donating the one-millionth HeartStart automated external ...
October 2, 2012 — A novel blood thinner recently approved by the U.S. Food and Drug Administration (FDA), Pradaxa ...
Apple created a stir when it announced in 2018 that its Apple Watch Series 4 was the first consumer health and fitness ...
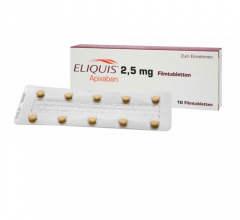
October 1, 2012 — Bristol-Myers Squibb Company and Pfizer Inc. announced that the U.S. Food and Drug Administration (FDA ...
September 26, 2012—The American College of Cardiology (ACC) has certified Lumedx software as fully interoperable between ...

September 24, 2012 — Surgically inserted cardiac devices play an important role in treating certain heart problems. In ...


 October 29, 2012
October 29, 2012