March 26, 2013 — According to two different reports from Millennium Research Group (MRG), the overall European market ...
Cath Lab
The catheterization lab channel includes content related to interventional technologies for coronary and peripheral artery disease (PAD). Other cath lab transcatheter device technologies covered on this page included percutaneous treatment for stroke, venous interventions, heart valves, hypertension, heart failure and percutenous coronary interventions (PCI).
March 26, 2013 — A multicenter analysis, led by Weill Cornell Medical College and published in the Journal of Clinical ...
March 25, 2013 — W. L. Gore & Associates introduced its Gore DrySeal Sheath with hydrophilic coating in Europe. The new ...
Adjustable parameters on the ACIST® CVI® Contrast Delivery System include, flow rate, volume, pressure limit, rise time ...

March 22, 2013 — Abbott announced that the U.S. Food and Drug Administration (FDA) Circulatory System Devices Panel of ...
March 21, 2013 — Cordis Corp. announced it has completed the acquisition of Flexible Stenting Solutions Inc. Currently ...
March 20, 2013 — Svelte Medical Systems announced treatment of the first patient in the DIRECT II (Direct Implantation ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...
March 20, 2013 — TherOx Inc. announced the initiation of its multicenter investigational device exemption (IDE) pilot cl ...
March 19, 2013 — Terumo Interventional Systems expanded its family of Heartrail III Coronary Guiding Catheters to ...
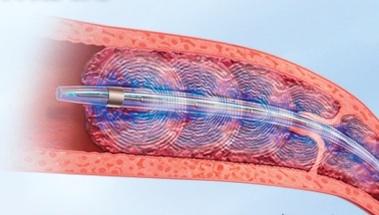
March 19, 2013 — Results of the ULTIMA trial using endovascular therapy to treat submassive pulmonary embolism (PE) were ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
Ajay Kirtane, M.D., SD, chief academic officer at Columbia University Medical Center/New York Presbyterian Hospital ...
The American College of Cardiology (ACC) 2013 Scientific Session Committee Chairman Miguel Quinones, M.D., explains some ...
DAIC Editor Dave Fornell explains his choices for the most innovative new technology on the expo floor at the American ...
During cardiology fellowship, my institution had a hand manifold system. I found it difficult to use at times, and it ...
March 18, 2013 – At the American College of Cardiology (ACC) conference, GE Healthcare unveiled DoseMap to alert ...
March 15, 2013 — Abbott announced positive long-term results for the company's innovative Absorb Bioresorbable Vascular ...
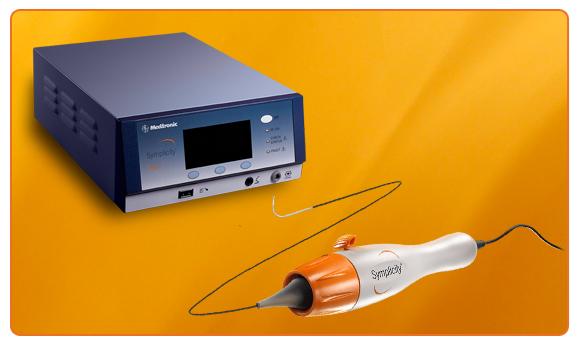
March 12, 2013 — Medtronic Inc. announced the U.S. Food and Drug Administration (FDA) and the Centers for Medicare and ...


 March 26, 2013
March 26, 2013









