Cath Lab
The catheterization lab channel includes content related to interventional technologies for coronary and peripheral artery disease (PAD). Other cath lab transcatheter device technologies covered on this page included percutaneous treatment for stroke, venous interventions, heart valves, hypertension, heart failure and percutenous coronary interventions (PCI).


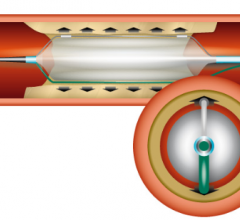
Adjustable parameters on the ACIST® CVI® Contrast Delivery System include, flow rate, volume, pressure limit, rise time ...

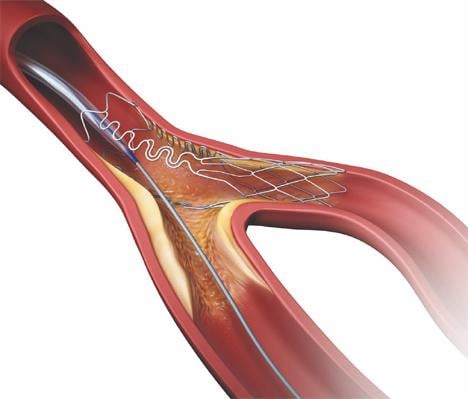
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...

October 30, 2013 — The first results from the CoreValve U.S. pivotal trial, the first U.S. data presented on the ...
October 30, 2013 — Terumo Interventional Systems announced the launch of the 0.018-inch Glidewire Advantage peripheral g ...
October 30, 2013 — Philips Healthcare and Infraredx Inc. announced a non-exclusive resale agreement for Infraredx’s TVC ...
During cardiology fellowship, my institution had a hand manifold system. I found it difficult to use at times, and it ...
October 29, 2013 — The findings of a Harvey L. Neiman Health Policy Institute study published online in the Journal of ...

 November 01, 2013
November 01, 2013