September 1, 2015 — Based on its recent analysis of the cardiovascular image management market, Frost & Sullivan ...
Cath Lab
The catheterization lab channel includes content related to interventional technologies for coronary and peripheral artery disease (PAD). Other cath lab transcatheter device technologies covered on this page included percutaneous treatment for stroke, venous interventions, heart valves, hypertension, heart failure and percutenous coronary interventions (PCI).
September 1, 2015 — A bioresorbable drug-eluting coronary stent showed similar efficacy and safety results compared to a ...
September 1, 2015 — Extending treatment with the anticoagulant bivalirudin for at least four hours after completion of ...
Adjustable parameters on the ACIST® CVI® Contrast Delivery System include, flow rate, volume, pressure limit, rise time ...
August 31, 2015 — Mitralign Inc. said the U.S. Food and Drug Administration (FDA) granted an investigational device ...
August 28, 2015 — PinnacleHealth CardioVascular Institute enrolled the first patient in Pennsylvania and second in the ...
August 28, 2015 — Philips Healthcare is showcasing its latest cardiology solutions at the European Society of Cardiology ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...
August 28, 2015 — BioControl Medical said it has completed enrollment in its INOVATE-HF (INcrease Of VAgal TonE in Heart ...
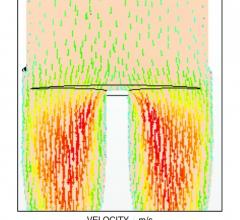
August 27, 2015 — The use of computational fluid dynamics (CFD) modeling has been used for years to better engineer ...
August 26, 2014 — The Cardiovascular Research Foundation (CRF) announced the late-breaking trials and first report ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...

August 25, 2015 — Medtronic announced it entered a definitive agreement to acquire Twelve Inc., a start–up medical ...
August 21, 2015 — CompView Medical announced the release of the NuCART, a turn-key, all-in-one mobile boom system with ...
August 21, 2015 — Loyola University Medical Center is the first and only hospital in Illinois to offer a new ...
During cardiology fellowship, my institution had a hand manifold system. I found it difficult to use at times, and it ...
August 20, 2015 — Boston Scientific Corp. has received U.S. Food and Drug Administration (FDA) approval for the Innova ...
August 19, 2015 – Corindus Vascular Robotics Inc. and Unfors RaySafe Inc., a Fluke Biomedical Company, announced a ...
August 18, 2015 — Sunshine Heart Inc. announced an update in late July on its COUNTER HF U.S. pivotal study for the C ...


 September 01, 2015
September 01, 2015