July 13, 2016 — A new study in JACC: Cardiovascular Interventions supports access through the wrist, or radial access ...
Cath Lab
The catheterization lab channel includes content related to interventional technologies for coronary and peripheral artery disease (PAD). Other cath lab transcatheter device technologies covered on this page included percutaneous treatment for stroke, venous interventions, heart valves, hypertension, heart failure and percutenous coronary interventions (PCI).
Angiographic imaging system vendors have developed new technologies to address emerging cath lab trends, including the ...
July 11, 2016 — University Hospitals Case Medical Center, Cleveland, is among the first in the country – and the first ...
Adjustable parameters on the ACIST® CVI® Contrast Delivery System include, flow rate, volume, pressure limit, rise time ...
July 8, 2016 — Kirk Schoenherr, 56, of Clinton Township, Mich., recently became just the second patient at Beaumont ...
July 5, 2016 — The U.S. Food and Drug Administration (FDA) has cleared the first fully bioresorbable coronary stent for ...
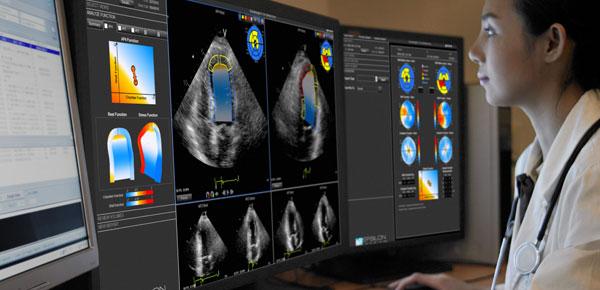
Below is a round up of news and videos about new cardiac ultrasound technology discussed in sessions and on the expo ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...
June 30, 2016 — Vital Images Inc. recently launched version 7 of its Vitrea advanced visualization software. This ...
June 30, 2016 — BioVentrix Inc. announced that it has received certification for CE marking its Revivent TC ...
Interview with Stephen Little, M.D., medical director of the Valve Clinic at the Houston Methodist DeBakey Heart and ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
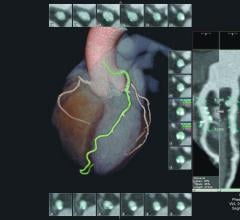
June 29, 2016 — HeartFlow Inc. announced that it is launching its next generation of the HeartFlow FFR-CT Analysis. The ...
Interview with Rebecca Hahn, M.D., FASE, Columbia University Medical Center, New York, at the American Society of ...
June 21, 2016 — Zoll Medical Corp. announced that patients experience a high one-year survival rate following use of the ...
During cardiology fellowship, my institution had a hand manifold system. I found it difficult to use at times, and it ...
This is an animation, supplied by Gore, demonstrated how the Cardioform Septal Occluder is implanted for the ...
I saw a new combination of technologies on the expo floor at the American Society of Echocardiography (ASE) meeting in ...
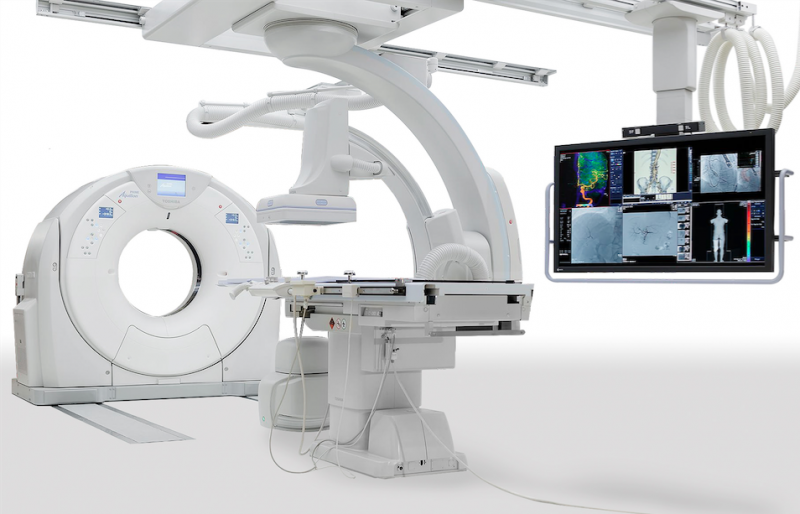
June 13, 2016 — GE Healthcare and Getinge Group announced the U.S. launch of a new, highly flexible angiography solution ...


 July 13, 2016
July 13, 2016