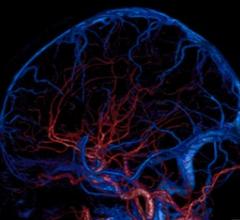
March 13, 2015 — Researchers at the University of North Carolina (UNC) School of Medicine have found the blood platelet ...
Antiplatelet and Anticoagulation Therapies
This channel includes news and new technology innovations for cardiovascular pharmaceutics. This includes antiplatelet agents, anticoagulation drugs, international normalized ratio (INR) testing, oral anticoagulants, IV administered drugs such as Heprin, and dual antiplatelet therapy (DAPT). The most commonly used anticoagulant is warfarin, which available in generic form at a low cost. However, it has a narrow therapeutic window and its effectiveness is altered by food containing vitamin K. To regulate warfarin, regular INR testing is needed. The newer anticoagulation drugs are referred to as novel oral anticoagulant (NOAC). However, since some of these drugs are now more than six years old, they are commonly referred to as non-vitamin K oral anticoagulant (NOAC), and the newest term, direct oral anticoagulant (DOAC). NOAC or DOAC agents have much larger therapeutic windows, do not require INR testing. Aspirin and clopidogrel (Plavix) are the most commonly used antiplatelet agents for the prevention of heart attacks and stroke. They are often prescribed together as DAPT.
March 11, 2015 — Point of Care Anticoagulation software (PCDS AC) optimizes delivery of anticoagulation (AC) drugs and ...
March 9, 2015 — Nearly all women and people over 65 in the United States with atrial fibrillation are advised to take bl ...
March 4, 2015 — The U.S. Food and Drug Administration (FDA) has approved the Xarelto Starter Pack for deep vein ...
February 26, 2015 — Patients who experienced a certain type of heart attack who received the anticoagulant fondaparinux ...
February 26, 2015 — By loading magnetic nanoparticles with drugs and dressing them in biochemical camouflage, Houston ...
January 22, 2015 — AstraZeneca announced that the PEGASUS-TIMI 54 study, a large-scale outcomes trial involving more ...
January 21, 2015 — A new post-marketing study evaluating the safety of once-daily rivaroxaban (Xarelto) shows, in ...
The America College of Cardiology has released its list of key late-breaking clinical trials at the ACC 2015 meeting in ...

January 14, 2015 — More than 10 percent of patients treated with aspirin therapy for primary cardiovascular disease ...
January 12, 2015 — Daiichi Sankyo Co. Ltd. announced that the U.S. Food and Drug Administration (FDA) has approved ...
January 7, 2015 — Discussions regarding late-breaking cardiovascular clinical trials among leading medical scientists ...
December 30, 2014 — Wearable technologies, possible changes to stenting practices, state legislation on energy drinks ...
December 23, 2014 — The most accessed research studies in the Journal of the American College of Cardiology (JACC) in ...
December 16, 2014 — Alere Inc. has initiated a voluntary correction to inform United States users of the Alere INRatio ...

 March 13, 2015
March 13, 2015