March 14, 2013 — Lantheus Medical Imaging Inc. announced the U.S. Food and Drug Administration (FDA) has granted approval of a supplemental new drug application (sNDA) that allows Jubilant HollisterStier (JHS) to be a new manufacturing site for its ultrasound imaging agent, Definity Vial for (Perflutren Lipid Microsphere) Injectable Suspension.

March 13, 2013 — St. Jude Medical Inc. announced the first patient implant in a new pivotal trial evaluating the company’s Amplatzer cardiac plug (ACP) for the prevention of stroke.
March 13, 2013 — CircuLite Inc. announced it has received conditional approval from the U.S. Food and Drug Administration (FDA) for an investigational device exemption (IDE) for its lead product, the Synergy circulatory support system, a minimally invasive device designed to reverse the symptoms of heart failure in ambulatory chronic heart failure patients.
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
http://www.thelancet.comAtherosclerosis is usually considered to be related to contemporary risk factors such as smoking, obesity and lack of exercise. However, researchers suggest that high prevalence of atherosclerosis in pre-modern humans may support the possibility of a more basic human predisposition to the disease.

Many physicians have long believed that the use of intravenous contrast agents for computed tomography (CT) scans can cause acute kidney injury. New Mayo Clinic research questions the strength of the causal link between the two. The findings from two tandem studies are published online in the journal Radiology.
Using a telemedicine system to engage people in underserved, urban communities to measure and report their blood pressure remotely — outside of the doctor’s office — appears to help them achieve blood pressure goals and improve adherence to lifestyle changes and medication recommendations. This is according to research being presented at the American College of Cardiology’s 62nd Annual Scientific Session. Overall, researchers say that just being in a system of care, with or without telemedicine, can result in important reductions in blood pressure.
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
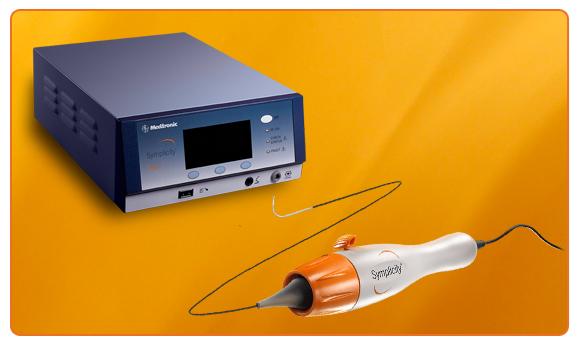
Medtronic Inc. announced the U.S. Food and Drug Administration (FDA) and the Centers for Medicare and Medicaid Services (CMS) have accepted the inclusion of the Symplicity renal denervation system for treatment-resistant hypertension in their parallel review program, which will allow CMS to begin consideration for national coverage determination while the FDA completes its review of safety and efficacy.
With the recent release of the updated American Heart Association (AHA)/American Stroke Association (ASA) Guidelines for Early Management of Acute Stroke Patients, Physio-Control announced that the LifeNet System’s OnePush feature can be used to rapidly activate stroke care teams. LifeNet’s stroke alert capability enables faster time-to-treatment, assisting stroke care teams in meeting the updated AHA/ASA Guidelines and helping improve stroke patient outcomes. The updated guidelines, released Jan. 31, 2013, support the overarching concept of stroke systems of care and detail aspects of stroke care from patient recognition; emergency medical services activation, transport and triage; through the initial hours in the emergency department and stroke unit.[1]
March 11, 2013 — HealthHelp announced it will launch in select markets in May its MedTree QDS, a transparent clinical decision support (CDS) system that allows providers to self-govern the ordering process and receive approval for a test or procedure in less than one minute.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
Royal Philips Electronics and Infraredx Inc. announced the availability of a new solution that enables seamless access to Infraredx’s true vessel characterization (TVC) Imaging System via Philips’ Allura Xper catheterization (cath) lab imaging systems. The product offering ensures compatibility between the two systems and allows clinicians to quickly and easily connect the TVC Imaging System to the Allura Xper system when intravascular imaging is required as part of cardiac catheterization procedures.

Even minor weight loss is associated with worse health outcomes among patients implanted with a certain type of defibrillator known as cardiac resynchronization therapy with defibrillator (CRT-D), according to research being presented at the American College of Cardiology’s 62nd Annual Scientific Session.
Lumedx unveiled a host of new HealthView products at the American College of Cardiology Scientific Sessions in San Francisco, March 9-11, 2013. New solutions include a comprehensive, Web-based CVIS with numerous newly released and upgraded components.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
Siemens Healthcare has announced that the U.S. Food and Drug Administration (FDA) has cleared its Artis Q and Artis Q.zen angiography system families, which feature revolutionary new X-ray tube and detector technology designed to improve minimally invasive therapy of diseases such as coronary artery disease (CAD), stroke and cancer. The new X-ray tube in both the Artis Q and Artis Q.zen can help physicians identify small vessels up to 70 percent better than conventional X-ray tube technology. The Artis Q.zen combines this X-ray source with a new detector technology that supports interventional imaging in ultra-low-dose ranges to patients, doctors and medical staff — particularly during longer interventions. With the Artis Q and Artis Q.zen system families, Siemens Healthcare again demonstrates its innovative strength and market competitiveness — key components of its Agenda 2013 two-year global Sector initiative.
As cardiologists assemble in San Francisco for the 2013 Scientific Sessions of the American College of Cardiology, they are faced with the challenge of how to handle the high volume of additional patients entering the healthcare system due to the Affordable Care Act.
Boston Scientific Corporation has begun the launch of its 0.035-inch and 0.018-inch Rubicon Support Catheter in the United States. The device is designed to assist physicians with placement and support of guidewires that are used in peripheral vascular procedures to deliver stents and balloons to open blockages in the legs and other peripheral arteries. Boston Scientific is also launching the 0.035-inch Rubicon Support Catheter in Europe to build on the momentum of the 0.018-inch size that was introduced in that region last fall.

 March 14, 2013
March 14, 2013