October 15, 2012 — Cardiac Science has unveiled its Burdick 4250 Holter recorder — a wearable heart monitor half the size and weight of its Holter monitoring system predecessor.
Three-year data from the Zilver PTX randomized controlled trial of paclitaxel-eluting stents for femoropopliteal disease from Cook Medical demonstrate 70.7 percent primary patency in the superficial femoral artery (SFA) at 36 months for patients treated with the paclitaxel-eluting stent.
Surefire Medical Inc. has received U.S. Food and Drug Administration (FDA) 510(k) clearance to market its line of Surefire angiographic catheters. Surefire Medical will launch these products in the United States later this year.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
October 12, 2012 — Biotronik announced the start of the prospective multicenter PARCADIA study. The study aims to identify risk factors that can help predict appropriate ICD interventions in patients with ischemic cardiomyopathy who have received an ICD for primary prevention according to the current ESC (European Society of Cardiology) guidelines.

October 12, 2012 — Boston Scientific announced it has enrolled the first patient in the REPRISE II clinical trial to evaluate the safety and performance of the Lotus valve system in up to 120 patients with severe aortic valve disease.
October 12, 2012 — Estech, a provider of minimally invasive cardiac ablation devices, last month announced the market release of its Cobra Fusion ablation system.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
TyRx Inc. announced that it has received a license from Health Canada to market its AigisRx Antibacterial Envelope. The AigisRx Envelope is specifically designed to stabilize pacemakers and implantable cardioverter defibrillators (ICD) while also releasing antimicrobial agents to help provide protection from microbial colonization of the device during surgical implantation.
October 11, 2012 — Spectranetics Corp. announced the U.S. market launch of the Tapas catheter at the 2012 Vascular Interventional Advances (VIVA) conference in Las Vegas. Tapas is manufactured by ThermopeutiX Inc. and distributed by Spectranetics.
October 11, 2012 — Edwards Lifesciences Corp. announced it has completed the acquisition of BMEYE B.V., a privately held Dutch company that specializes in the development of noninvasive technology for advanced hemodynamic monitoring.
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
October 11, 2012 — With this month’s approval of the Standards and Guidelines for Advanced Cardiovascular Sonography by the Commission on Accreditation for Allied Health Education Programs (CAAHEP), experienced cardiovascular sonographers have a new educational pathway for career advancement.
Boston Scientific Corporation has begun the United States and European launch of its Victory guidewire, designed to facilitate crossing of resistant lesions and the placement and exchange of balloon catheters or other interventional devices within the peripheral vasculature. The company expects to launch the product in other international markets later this year and in 2013, subject to regulatory approvals.
New research from the Netherlands suggests tiny microbubbles can be used to more effectively deliver a blood clot busting drug to patients while they are in the ambulance during acute heart attack.
Change Healthcare Cardiology Hemodynamics is an integrated hemodynamic monitoring system for monitoring vital signs and ...
The U.S. Food and Drug Administration (FDA) has granted Boston Scientific Corp. regulatory approval for its S-ICD System, the world's first commercially available subcutaneous implantable cardioverter defibrillator (S-ICD). It sits entirely just below the skin without the need for implantable lead to be placed inside the heart.
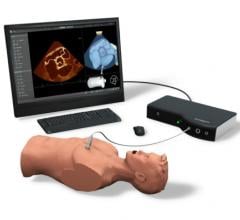
October 8, 2012 — Simbionix USA Corp. will introduce the new advanced U/S (ultrasound) Mentor simulator at the upcoming American College of Emergency Physicians (ACEP) Exhibition in Denver, Colo. This new Simbionix simulator offers realistic hands-on training for diverse ultrasound examinations and interventions.
Following Health Canada approval, Cook Medical has made the Zilver Vena Venous Self-Expanding Stent available to physicians across Canada. Designed to restore blood flow in obstructed iliofemoral veins, Zilver Vena provides physicians with a tool designed specifically for stenting obstructed iliofemoral veins.


 October 15, 2012
October 15, 2012