Welch Allyn has been working with Epic to connect several of its vital signs devices, monitors and electrocardiograph (ECG) units to Epic’s electronic medical records (EMR) system to help eliminate the need for manual data entry by providers, increase efficiency and alleviate the risk of transcription and latency errors. By establishing connectivity between Welch Allyn devices and the EMR, clinicians are able to transfer patient test results directly between diagnostic devices and the EMR — resulting in immediate access to accurate patient data and improved workflow.
Cerebral function is the essence of quality of life. Its preservation throughout medical procedures is a key component to procedural success and patient care. New scientific data presented at the annual EuroPCR Congress in Paris heightens the spreading understanding and recognition that protecting the brain and preserving brain reserve is an important goal of TAVR and other cardiovascular procedures.
Kona Medical announced three and six month results from the WAVE I study, a first-in-man study evaluating the safety and efficacy of Kona Medical’s Surround Sound Renal Denervation System for the treatment of resistant hypertension.
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
CardioKinetix Inc. announced the transmission of a live satellite feed of a clinical case using the catheter-based Parachute Device. The Parachute procedure was performed by Dr. Martyn Thomas, M.D., chairman of cardiology at St. Thomas’ Hospital in London, England, with assistance from Dr. Hüseyin Ince, professor of medicine, University Hospital Rostock, Germany.

When a power outage occurs, MRI equipment can be damaged and accuracy of the scans effected. To mitigate the effects of power interruptions, healthcare facilities have traditionally relied on the lead-acid batteries of uninterruptible power supplies (UPSs) to provide backup power. However, batteries are notorious for their reliability issues, large footprint and frequent maintenance and replacement.

Today’s healthcare is changing in many ways and so are the processes, technology and people delivering the extremely complex cardiology procedures and services. There are multiple biomedical and technical advances that are proving to optimize patient care. With declining budgets and reimbursements, healthcare providers are under increasing pressure to develop effective ways of improving efficiency and reducing costs, while maintaining high levels of patient care.
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...

Cardiovascular Systems Inc. (CSI) has presented 30-day results from its ORBIT II study of coronary artery disease. ORBIT II is evaluating the safety and effectiveness of the company’s orbital atherectomy technology in treating patients with severely calcified coronary lesions. This is the first Investigational Device Exemption study to evaluate this problematic subset of patients. CSI completed ORBIT II enrollment of 443 patients at 49 U.S. medical centers in November 2012 and submitted its Premarket Approval application to the U.S. Food and Drug Administration (FDA) on March 15, 2013.
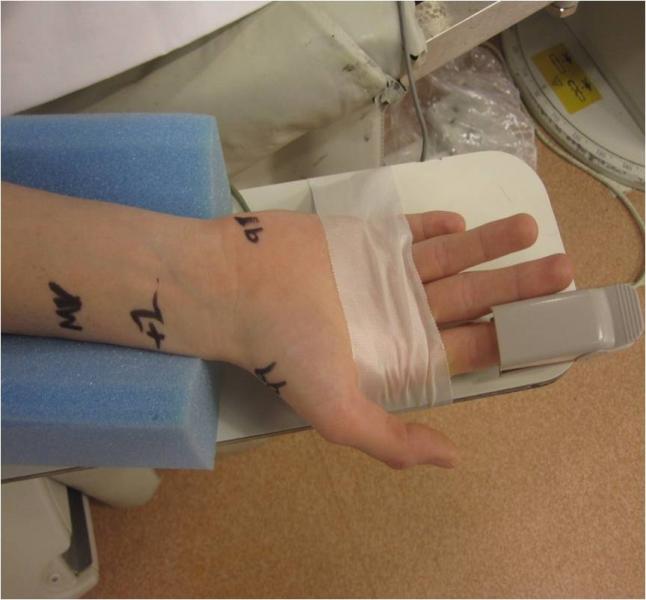
Transradial coronary artery angiography was a major focus at the Society for Cardiovascular Angiography Interventions (SCAI) 2013 Scientific Sessions May 8-11 in Orlando, Fla. The meeting kicked-off with a dedicated symposium covering topics such as access, radiation exposure and complications.
Philips Healthcare announced that CX50 xMatrix, the first portable ultrasound with Philips' Live 3-D transesophageal echo (TEE), now offers 2-D intracardiac echo (ICE) capability. The CX50 xMatrix with available Live 3-D TEE and ICE was first shown in Paris at the EuroPCR in May.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
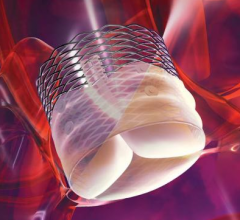
The Lotus Valve, a second-generation transcatheter aortic valve implantation (TAVI) device, was successfully implanted in all of the first 60 patients in results from REPRISE II, which showed good device performance and low mortality at 30 days.
The U.S. District Court for the District of New Jersey approved an agreement under which Vascular Solutions Inc. will cease further manufacture, sales and distribution of its R-Band Radial Hemostasis Device, pending litigation of Terumo Medical's patent infringement claims.
Direct Flow Medical Inc. said it met its primary endpoint in the fully-enrolled, 100-patient DISCOVER CE mark trial by achieving 99 percent freedom from all-cause mortality at 30 days. Patients were treated with the Direct Flow Medical Transcatheter Aortic Valve System and demonstrated excellent results relative to overall patient hemodynamics, as well as the avoidance of post-procedural aortic regurgitation and adverse events. The DISCOVER Trial full 30-day outcomes were presented today by trial investigator Joachim Schofer, M.D., from the Medical Care Center, Hamburg, Germany.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
Abbott announced CE mark in Europe for the Xience Xpedition 48 Everolimus Eluting Coronary Stent System, the first-of-its-kind treatment for very long blockages in the vessels that supply blood to the heart due to coronary artery disease (CAD).
CardioKinetix Inc. announced results of a meta-analysis study of the catheter-based Parachute Ventricular Partitioning Device. Six-month clinical results from 91 U.S. and European patients with ischemic heart failure were presented.

InspireMD Inc. announced new six-month results from the MASTER trial demonstrating that the MGuard embolic protection stent (EPS) outperformed bare metal stents and drug-eluting stents in all-cause mortality in ST-segment elevation myocardial infarction (STEMI) patients.


 May 30, 2013
May 30, 2013