Amaranth Medical announced that it completed enrollment in May in the RENASCENT-II study of its novel Aptitude 120-micron sirolimus-eluting bioresorbable scaffold (BRS).
Biotronik announced results establishing non-inferiority of the Orsiro hybrid drug-eluting stent (DES) to the Resolute Integrity DES were presented during a Hotline Session at EuroPCR 2016.
June 3, 2016 — Cardionovum GmbH recently announced the completion of enrollment of the RAPID trial. Results will be used ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
According to data from a new survey, nearly all of the nation’s hospitals have adopted certified electronic health records (EHRs). The survey was released at the Office of the National Coordinator for Health Information Technology’s (ONC) 2016 annual meeting, May 31-June 2 in Washington, D.C.
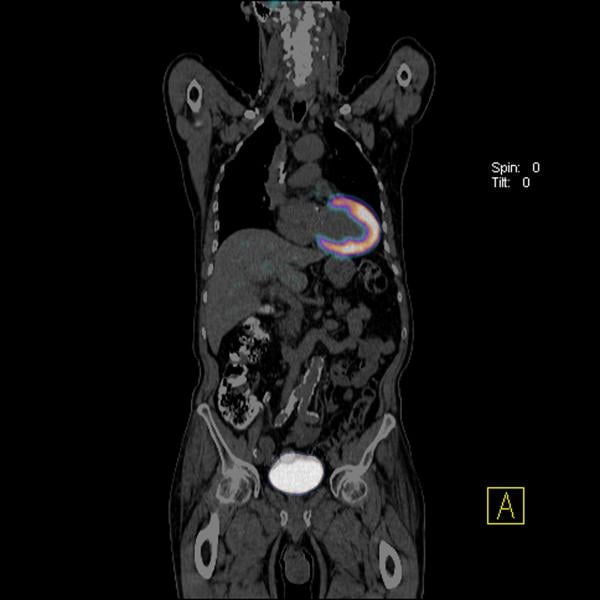
Positron emission tomography (PET) is a nuclear imaging technology (also referred to as molecular imaging) that enables ...
Female atrial fibrillation patients are less likely than their male counterparts to receive blood thinning therapies to prevent stroke, say University of Cincinnati College of Medicine researchers.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
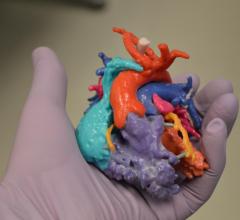
June 2, 2016 — Artec 3D, a developer and manufacturer of professional 3-D hardware and software, announced the release ...
June 2, 2016 — Here are the top 20 most popular current articles, news releases and videos on the Diagnostic and ...

Healthcare reform efforts of recent years have challenged the industry to reduce the costs of healthcare for both ...
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
The American Society of Echocardiography (ASE) will host its 27th Annual Scientific Sessions, June 10-14, 2016, at the Washington State Convention Center in Seattle.
BioVentrix Inc. announced that it has received a U.S. Food and Drug Administration (FDA) Investigational Device Exemption (IDE) approval to initiate its pivotal clinical trial, named ALIVE (American Less Invasive Ventricular Enhancement).
Biotronik announced the launch of CardioMessenger Smart in the United States. CardioMessenger Smart is a portable monitoring device, about the size of a modern smartphone, that keeps pacemaker, implantable cardioverter defibrillator (ICD) and insertable cardiac monitor (ICM) patients connected to their physician remotely, enabling more efficient care management anywhere in the world.
Change Healthcare Cardiology Hemodynamics is an integrated hemodynamic monitoring system for monitoring vital signs and ...
A person is admitted to the hospital with a stroke, but not much is known about whether or not that patient will undergo neuroimaging.
In a recent study, the use of a stent to repair pulmonary artery stenosis in children and adults with congenital heart disease was successful in the majority of patients, but many also experienced serious complications. The study was published in March in the Journal of the American College of Cardiology.
MIM Software Inc. announced it has received 510(k) U.S. Food and Drug Administration (FDA) clearance to market full MIM products running on tablets through thin client technology such as Citrix.

 June 06, 2016
June 06, 2016