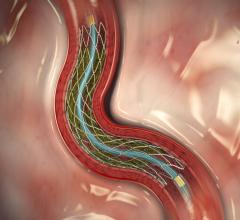
Studies have shown transcatheter aortic valve replacement (TAVR) has an increased risk of stroke and cerebral damage due ...
Edwards Lifesciences Corp. recently announced new data demonstrating dramatic and sustained improvements in quality of life for severe aortic stenosis (AS) patients at intermediate surgical risk treated with Edwards transcatheter heart valves. Study results were presented at the 28th Transcatheter Cardiovascular Therapeutics (TCT), the annual scientific symposium of the Cardiovascular Research Foundation, in Washington, D.C.

November 7, 2016 – Results from the U.S. real-world, post-FDA approval experience of the Watchman device found high ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
Keystone Heart Ltd. announced that data presented at the Transcatheter Cardiovascular Therapeutics (TCT) 2016 scientific symposium demonstrate an increase in brain lesions for patients following transcatheter aortic valve replacement (TAVR) procedures, which were significantly associated with delirium.
November 7, 2016 – Results from TRANSFORM-OCT, a prospective, randomized trial using optical coherence tomography (OCT) ...
November 7, 2016 – A multicenter randomized trial evaluating the role of embolic protection using the Sentinel device ...
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
November 7, 2016 – The two-year results from LEADERS FREE, the first randomized clinical trial dedicated to high ...
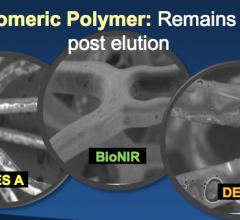
The 28th annual Transcatheter Cardiovascular Therapeutics (TCT) scientific symposium, Oct. 29-Nov. 3 in Washington, D.C., featured a number of first report investigations on novel stents that could become the next generation of bioresorbable stents in patients.
November 7, 2016 – The large multinational randomized BIONICS study found that a novel elastic polymer coated ...
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
November 7, 2016 – Two-year results from the COLOR Trial, the first large-scale multicenter prospective study of its ...

November 4, 2016 — Here is the list of the top 20 most popular pieces of content on the Diagnostic and Interventional ...
Coronary artery bypass (CABG) surgery is the standard treatment for revascularization in patients with left main coronary artery (LMCA) disease, but use of percutaneous coronary intervention (PCI) for this indication is increasing. Findings from the Nordic–Baltic–British Left Main Revascularization Study (NOBLE) trial found that despite similar mortality, the five-year risk of major adverse events was higher after PCI compared to CABG for the treatment of unprotected LMCA disease.
Change Healthcare Cardiology Hemodynamics is an integrated hemodynamic monitoring system for monitoring vital signs and ...
St. Jude Medical Inc. announced the long-term data from RESPECT, a landmark trial, during a First Report session at the 2016 Transcatheter Cardiovascular Therapeutics (TCT) conference in Washington, D.C.
Investigators recently unveiled clinical data from the independent BIO-RESORT study, representing the first all-comers analysis to compare the safety and efficacy of biodegradable polymer stents (BP-DES) to the durable polymer Resolute Integrity drug-eluting stent (DP-DES) from Medtronic. At one year, patients with coronary artery disease who were treated with a biodegradable polymer stent showed no clinical benefits over patients treated with Resolute Integrity.
Medtronic plc recently announced new clinical data for the Harmony Transcatheter Pulmonary Valve (TPV) from its early feasibility study, showing improved hemodynamics from baseline and consistent valve performance at one year. Presented at the Transcatheter Cardiovascular Therapeutics (TCT) scientific symposium, the positive new data from the first-of-its-kind early feasibility study led the U.S. Food and Drug Administration (FDA) to also recently approve the initiation of a Pivotal Investigational Device Exemption (IDE) study to evaluate the valve’s safety and effectiveness.

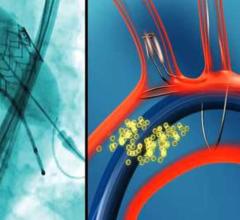
 November 08, 2016
November 08, 2016