Abiomed Inc. announced that it received an expanded U.S. Food and Drug Administration (FDA) Pre-Market Approval (PMA) for its Impella 2.5, Impella CP, Impella 5.0 and Impella LD heart pumps to provide treatment for heart failure associated with cardiomyopathy leading to cardiogenic shock. This approval expands the previous FDA indication for acute myocardial infarction (AMI) cardiogenic shock and post-cardiotomy cardiogenic shock (PCCS), received in April 2016.
February 15, 2018 — The National Institutes of Health (NIH) awarded a $2.2 million research grant to healthcare ...

February 15, 2018 — Edwards Lifesciences Corp. has received European CE mark clearance for its self-expanding Centera ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
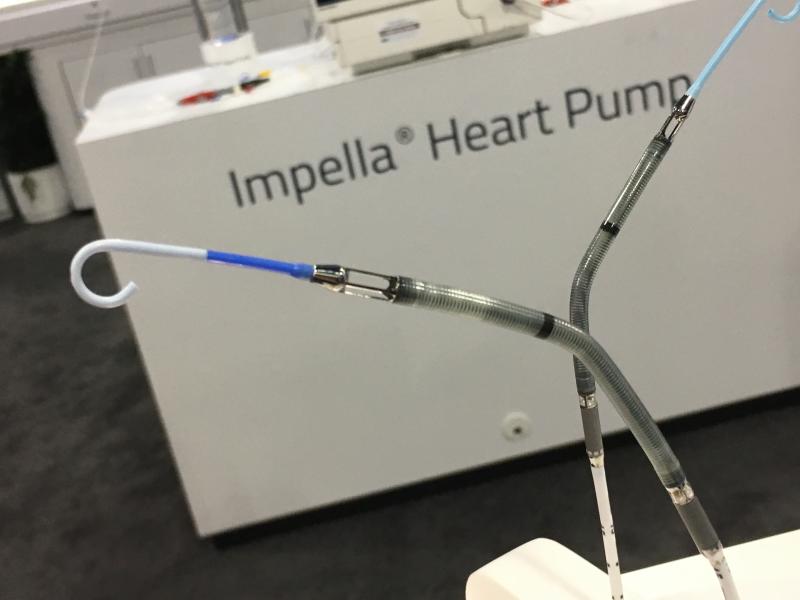
February 14, 2018 — Abiomed Inc. announced it received an expanded U.S. Food and Drug Administration (FDA) pre-market ...
February 14, 2018 — The U.S. Food and Drug Administration (FDA) announced marketing clearance for Viz.AI’s Contact ...

There have been several advancements in pacemaker technologies over the past few years. This is an overview of some of ...
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
Abbott announced the first patient has been enrolled in a clinical trial evaluating 28 days of dual antiplatelet therapy (DAPT) in patients at high risk of bleeding after implantation with a Xience everolimus-eluting coronary stent. The first patient was enrolled into the study by Prof. Emanuele Barbato, M.D., Ph.D., a cardiologist at OLV-Hospital Aalst in Belgium.

Clinical research has revealed men and women often have different presentations for cardiovascular disease (CVD). This ...
February 12, 2018 — Philips is recalling the HeartStart MRx Defibrillator due to a defect in the device's Gas Discharge ...
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
February 12, 2018 — Physicians now have access to more neuro and cardiac magnetic resonance imaging (MRI) capabilities ...
Fox Business News recently featured GE Healthcare’s ultrasound technologies in a 6-minute segment with the Innovations ...
The Instadose + dosimeter, the latest by Mirion Technologies, transforms radiation monitoring programs to increase ...
Change Healthcare Cardiology Hemodynamics is an integrated hemodynamic monitoring system for monitoring vital signs and ...
February 8, 2018 – Calgary Scientific announced a renewed focus on an enterprise cloud strategy, which will support its ...
February 8, 2018 – ScImage, a leading provider of enterprise PACS solutions, is celebrating 25 years of providing ...
Medtronic plc recently added to its body of clinical evidence supporting the In.Pact Admiral drug-coated balloon (DCB) with new presentations that demonstrated durable and consistent clinical outcomes in peripheral artery disease (PAD). The new data, presented at the Leipzig Interventional Course (LINC), Jan. 30-Feb. 2, 2018, in Leipzig, Germany, included the two-year results from the MDT-2113 study (IN.PACT SFA Japan) and data from a critical limb ischemia (CLI) subgroup analysis from the IN.PACT Global Study.


 February 15, 2018
February 15, 2018