Cerebrovascular imaging analysis company iSchemaView received final clearance from the U.S. Food and Drug Administration (FDA) for Rapid CTA, the company’s 3-D imaging solution for computed tomography angiography. Rapid CTA is the newest addition to iSchemaView’s neuroimaging platform — including Rapid CTP and Rapid MRI — which is designed to provide physicians with fast, fully-automated and easy-to-interpret imaging that facilitates clinical decision making around cerebrovascular disease, such as stroke.
Edwards Lifesciences Corp.announced that it received CE Mark for the Edwards Cardioband Tricuspid Valve Reconstruction System for the treatment of tricuspid regurgitation. The Cardioband Tricuspid System is the first commercially available transcatheter therapy for the treatment of tricuspid heart valve disease.

Here is the list of the most popular articles and videos on the Diagnostic and Interventional Cardiology (DAIC) magazine ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
April 30, 2018 — LivaNova announced it completed the sale of its cardiac rhythm management (CRM) business to MicroPort ...
Mandatory public reporting of coronary artery bypass grafting (CABG) results in Massachusetts was associated with better patient outcomes compared to national findings, according to a recent study. Results of the 13-year Massachusetts experience were presented in a plenary session of the American Association for Thoracic Surgery’s 98th Annual Meeting.

The introduction of thermal ablation revolutionized the treatment of varicose veins, yet recurrence remains a stubborn ...
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
The acquisition of biomedical equipment company Esaote SpA’s share capital was completed on April 19, the company recently announced, by a consortium of leading Chinese investors. The consortium is composed of major companies in the medical and healthcare technology sectors, as well as investment funds with significant experience in this field.
Point of Care Decision Support (PCDS) announced the release of a new version 2.0 of their anticoagulation clinical decision support (CDS) software. The application was developed with leading clinical thrombosis experts to specifically address the new and complex challenges of managing both warfarin and non-vitamin K antagonist oral anticoagulants (NOACs). It enables healthcare provider teams to deliver consistent, measurable and evidence-based quality of care for patients on anticoagulants.
ITN Associate Editor Jeff Zagoudis explores how the mobile stroke unit (MSU) program at Northwestern Medicine Central ...
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
Physio-Control, now part of Stryker, announced its newest version of the LUCAS 3 Chest Compression System, version 3.1. The latest version provides new capabilities for tailored device functionality, wireless reporting and device status notifications sent over email.
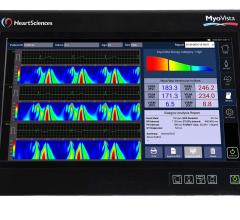
HeartSciences announced the results of a clinical study of its electrocardiography device that applies continuous wavelet transform signal processing and artificial intelligence. The study was serially conducted at The Icahn School of Medicine, Mount Sinai Hospital, New York, N.Y., and the West Virginia University (WVU) Heart and Vascular Institute, Morgantown, W. Va. The results were published online and in the April 17, 2018 issue of Journal of the American College of Cardiology (JACC).
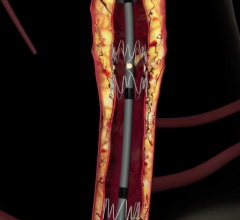
April 27, 2018 — Intact Vascular Inc. recently closed a Series C financing totaling $20 million. This financing is ...
Change Healthcare Cardiology Hemodynamics is an integrated hemodynamic monitoring system for monitoring vital signs and ...
The European Patent Office (EPO) has revoked Edward Lifesciences’ Corp.’s European patent EP 2,399,550 (‘550), ruling in favor of Boston Scientific and several other companies involved in an ongoing dispute related to transcatheter heart valves. The ‘550 patent is also the sole basis of Edwards’ infringement action against Boston Scientific in Ireland.

Over the past year, cardiac ultrasound vendors have introduced several new products to speed workflow, improve image ...

There have been a few big, recent advancements in cardiac computed tomography angiography (CCTA) imaging technology. The ...


 May 01, 2018
May 01, 2018