Amarin Corp. plc announced that its supplemental new drug application (sNDA) for Vascepa (icosapent ethyl) capsules has been accepted for filing and granted Priority Review designation by the U.S. Food and Drug Administration (FDA). The Prescription Drug User Fee Act (PDUFA) goal date assigned by the FDA for this sNDA is Sept. 28, 2019. Because of the Priority Review designation, the timing of this PDUFA date is four months earlier than the anticipated standard ten-month review for applications.
Philips announced the three-year results from the ILLUMENATE Pivotal trial and the ILLUMENATE European randomized clinical trial (EU RCT). These two trials are part of a series of five trials evaluating the safety and efficacy of Stellarex .035″ low-dose1 drug-coated balloon (DCB) to restore and maintain blood flow in the superficial femoral artery and popliteal arteries of patients with peripheral arterial disease. The results were evaluated compared to treatment with uncoated balloons, the current standard of care.
As part of the Consolidated Appropriations Act of 2018, pass-through payment status for LUMASON® (sulfur hexafluoride ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
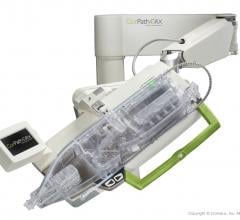
Corindus Vascular Robotics Inc. announced its CorPath GRX System was successfully used to perform a live complex robotic-assisted percutaneous coronary intervention (PCI) at the EuroPCR 2019 Conference, May 21-24 in Paris, France.
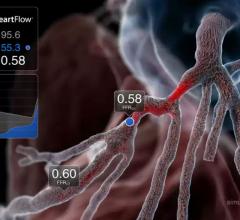
New data demonstrated that use of the investigational HeartFlow Planner, a real-time, non-invasive interactive planning tool, led to a change in treatment strategy in 45 percent of patients with coronary artery disease (CAD) and reduced the need for invasive physiology. The data from the BOWIE (Benefits of Obtaining information for planning With noninvasive FFRCT prior to Invasive Evaluation) study were presented by Eric Van Belle, M.D., Ph.D., professor of cardiology, head of the Lille Heart & Lung Institute, and principal investigator for BOWIE, as a late-breaking trial at the EuroPCR Conference, May 21-24 in Paris, France. The results were also published in the Journal of the American College of Cardiology (JACC).
Physicians use many strategies to better interface with patients and their families to try and explain in non-physician ...
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
Researchers have developed the first algorithm that can locate patient-specific ablation targets for atrial fibrillation (AFib) within the atria that does not require specialized catheters or 3-D electro-anatomic maps of the heart. The new algorithm – the iterative catheter navigation (ICAN) – is fundamentally different from existing approaches.

This is a brief overview of updates on implantable cardioverter defibrillators (ICD), including new technology ...
People who skip breakfast and eat dinner near bedtime have worse outcomes after a heart attack, according to research published in the European Journal of Preventive Cardiology,1 a journal of the European Society of Cardiology (ESC). The study found that people with both eating habits had a four to five times higher likelihood of death, another heart attack or angina (chest pain) within 30 days after hospital discharge for heart attack.
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
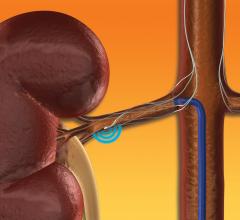
Investigators unveiled late-breaking clinical data from a first-of-its-kind physician sponsored clinical trial at the 2019 EuroPCR Annual Meeting, May 20-23 in Paris, France. The data indicate that renal denervation (RDN) with the Medtronic Symplicity renal denervation system was associated with reduced occurrence of subclinical atrial fibrillation (AF) in a small subset of high-risk patients with hypertensive heart disease over a median follow-up period of more than two years.
ControlRad Inc. announced that the U.S. Food and Drug Administration (FDA) granted 510(k) clearance for its ControlRad Trace and the company has initiated its commercial launch. The ControlRad Trace is the only technology, according to the company, that can be integrated into existing mobile C-arms to reduce radiation in any fluoroscopic imaging procedure.
Researchers at the National, Heart, Lung, and Blood Institute (NHLBI), part of the National Institutes of Health (NIH) ...
Change Healthcare Cardiology Hemodynamics is an integrated hemodynamic monitoring system for monitoring vital signs and ...
Abiomed announced that the Impella CP with SmartAssist will be commercially available beginning at the 2019 Society for Cardiovascular Angiography & Interventions (SCAI) Scientific Sessions, May 19-22 in Las Vegas, through a controlled launch process at select sites. The majority of Impella CP heart pumps in the U.S. will be transitioned to SmartAssist over the next fiscal year.
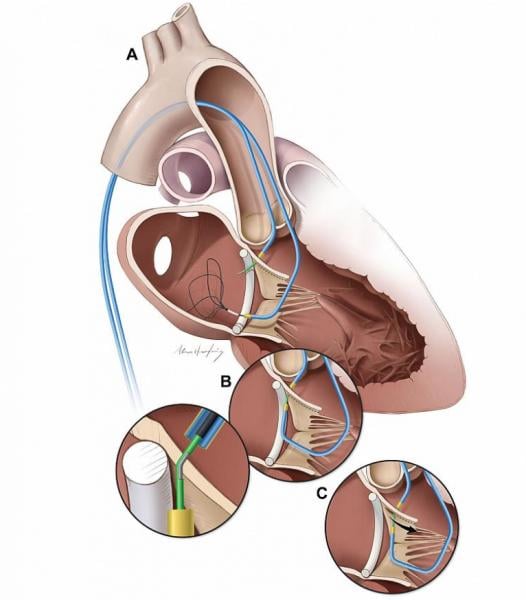
Edwards Lifesciences Corp. announced strategic clinical and regulatory milestones for its Edwards Pascal transcatheter valve repair system at the EuroPCR annual course, May 20-23 in Paris, France. New 6-month data from the CLASP study of the Pascal system were presented by Konstantinos Spargias, M.D., from the Hygeia Hospital in Athens, Greece. In addition, the U.S. Food and Drug Administration (FDA) has approved CLASP IIF, a prospective, multicenter, randomized, controlled pivotal trial studying the PASCAL system.
Medtronic plc announced its entrance into the guide extension catheter market with the global launch of the Telescope Guide Extension Catheter. The newly designed catheter is used to provide additional backup support and access to distal lesions. Guide extension catheters help deliver coronary stents, balloons and other interventional devices during angioplasty procedures that help to restore blood flow through the coronary and peripheral arteries.


 May 29, 2019
May 29, 2019