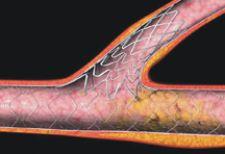
iffering approaches for the treatment of coronary bifurcation and ostial lesions (CBOL) continue to feed the debate over which stent designs and stenting techniques are most effective.
September 6, 2007 — ProSolv CardioVascular, a FUJIFILM company (ProSolv), announced at the 12th Annual Scientific ...
Just when drug-eluting stents (DES) were going to revolutionize interventional cardiology, new reports that DES may be associated with increased risk of late-stent thrombosis have many physicians putting the brakes on DES use. Despite this significant bump in the road, stent manufacturers are forging ahead with clinical trials on novel systems and on plans to roll out new DES platforms in 2008.
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
September 10, 2007 - Signalife launched the sales of its new, FDA-approved, ambulatory 12-lead ECG cardiovascular ...
According to the Centers for Disease Control and Prevention, more than 70 million Americans currently live with ...
September 10, 2007 - Four analyses of three-year follow-up data that were presented at the ESC Congress 2007 showed that ...
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
Possis Medical Inc. will feature the newest advancements in its “Complete Solution for Thrombus,” including the AngioJet Ultra Thrombectomy System, AngioJet Spiroflex and Spiroflex VG catheters and its Fetch Aspiration Catheter.
Combining all features of the ACIST CMS and Voyager systems onto one platform, the ACIST CVi Contrast Delivery System ...
Sony Electronics recently introduced its LMD-2450MD 24-inch monitor, which offers 10-bit signal processing and Sony ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
St. Jude Medical Inc. announced that the FDA cleared the ACross Transseptal Access System, a device designed to improve control and simplify the procedure physicians use to access the left atrium during complex cardiac procedures such as atrial fibrillation ablation.
September 5, 2007 – According to Boston Scientific Corp., results from the TAXUS WOMAN study, a gender specific analysis ...
GE Healthcare has released the latest version of Centricity Enterprise, an intelligent and integrated software suite of clinical and financial applications that is designed to improve patient care and increase collaboration between clinicians, providing a better overall healthcare environment.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
Siemens Automation and Drives (A&D) has added a 1MP color display to its portfolio of medical monitors. The SCD 19110 ...
September 6, 2007 – Boston Scientific Corp. announced at the ESC 2007 conference in Vienna, Austria, that results from ...
Cordis Endovascular announced, at the 61st Annual Meeting of the Society for Vascular Surgery, the worldwide launch of ...

 September 09, 2007
September 09, 2007











