January 26, 2016 — Fujifilm SonoSite Inc. announced CE mark and U.S. Food and Drug Administration (FDA) 510(k) clearance ...
Now, medical technology purchasers and users have an instant online forum to rate and review technologies they are using, with Rate This Model, the newest enhancement to the ECRI Institute’s SELECTplus capital decision support solutions suite.
St. Jude Medical is recalling the Optisure implantable cardioverter defibrillator (ICD) leads due to a manufacturing error that may have caused damage to the insulation layer of one of the shock coils. Depending on device programming and the depth of the cut, this could result in the inability of the defibrillator to deliver electrical therapy to the patient. The use of affected products may cause serious adverse health consequences, including patient injury or death.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
The suspension of the medical device excise tax will have positive consequences for the U.S. medical device market over the next few years, says an analyst with research and consulting firm GlobalData.
New recommendations from the American College of Cardiology (ACC) and the American College of Radiology (ACR) have established appropriate use of diagnostic imaging for patients with chest pain, one of the most common reasons for emergency department visits.
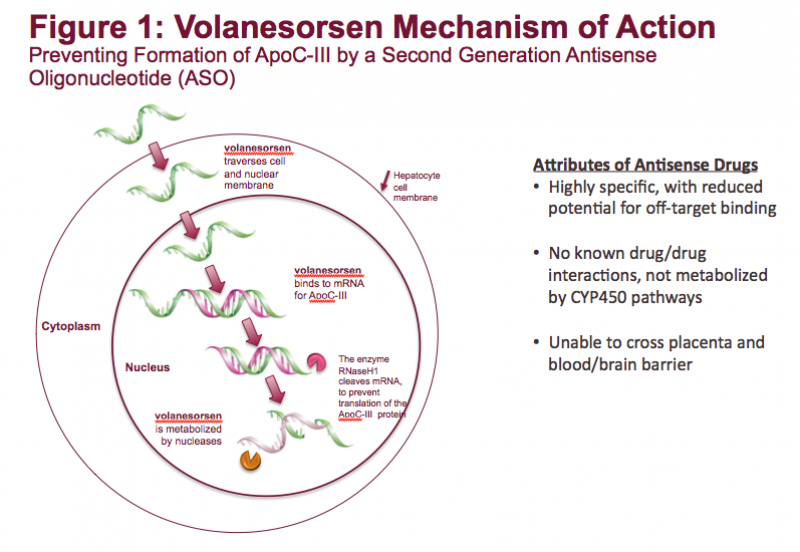
Familial chylomicronemia syndrome (FCS) is a rare, genetic disease characterized by mutations in genes affecting the ...
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
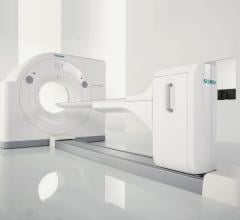
Siemens Healthcare has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the Biograph Horizon positron emission tomography/computed tomography (PET/CT) system, which offers premium performance at a low total cost of ownership.
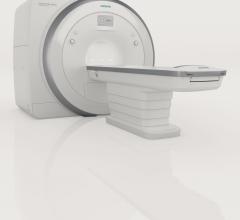
Siemens Healthcare announced that the U.S. Food and Drug Administration (FDA) has cleared the Magnetom Amira 1.5 Tesla magnetic resonance imaging (MRI) system.
This case study is from the Cardiac Imaging Department, Hospital Clinic, Barcelona, Spain. A woman in her early 60s came ...
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
GE Healthcare is taking the next leap in image quality performance, quantification and workflow with the introduction of ...
On the 100th anniversary of the Endurance expedition to Antarctica led by Sir Ernest Shackleton, doctors writing in the Journal of the Royal Society of Medicine believe the explorer may have had the congenital defect commonly known as a 'hole in the heart'.
Patients enrolled in high-deductible health insurance plans have lower rates of use and lower costs for imaging tests, suggests a study in the February issue of Medical Care.
Change Healthcare Cardiology Hemodynamics is an integrated hemodynamic monitoring system for monitoring vital signs and ...
A lengthy review of increasing medical evidence shows that cancer treatments such as chemotherapy and radiation often damage heart vessels and tissues, increasing the risk of later heart disease in cancer survivors. Now an interventional cardiologist from Detroit Medical Center (DMC) who is a co-author of the study is helping efforts to expand a new medical specialty known as “cardioncology.”
The U.S. Food and Drug Administration (FDA) Circulatory System Devices Panel is set to review data and offer recommendations concerning the final approval of Abbott’s Absorb fully bioresorbable stent and the AngelMed Guardian System.
A group of researchers from Russia, Australia and the Netherlands have developed a technology that can reduce magnetic resonance imaging (MRI) scanning times by more than 50 percent.


 January 26, 2016
January 26, 2016