July 8, 2020 – Philips Healthcare and Leeds Teaching Hospitals NHS Trust announced a seven-year managed service ...
July 7, 2020 — In the face of the coronavirus pandemic, public health officials worldwide are working tirelessly to slow ...
July 7, 2020 – MedAlliance announced enrollment of the first patient in its study of Selution SLR 0.014 drug-eluting ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...

Fractional flow reserve (FFR) pressure wires have been used now in interventional cardiology procedures for more than a ...
July 7, 2020 – Medtronic announced it received U.S. Food and Drug Administration (FDA) clearance and European CE mark ...
Canon Medical Systems USA has created a virtual trade-show experience for its cardiovascular computed tomography (CT) ...
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
July 6, 2020 — Black patients who undergo percutaneous coronary intervention (PCI) are at an increased risk for major ...
July 6, 2020 — Here is the list of the most popular content on the Diagnostic and Interventional Cardiology (DAIC) ...
July 1, 2020 — Carag AG announced receiving U.S. Food and Drug Administration (FDA) Investigational Device Exemption ...
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
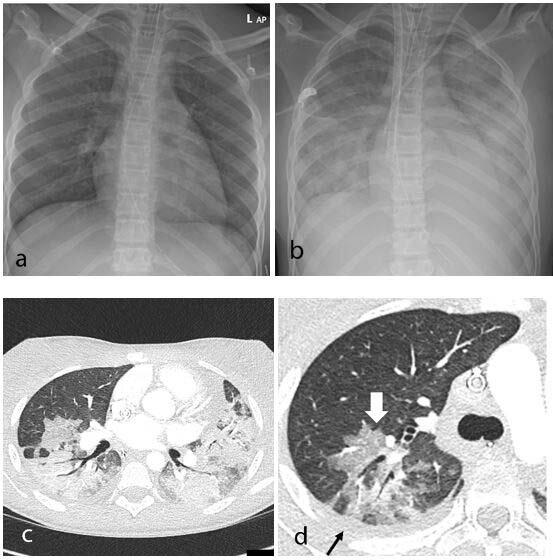
In recent weeks, a multisystem hyperinflammatory condition has emerged in children in association with prior exposure or ...
June 30, 2020 — DiA Imaging Analysis, a leading provider of advanced AI-based solutions for ultrasound analysis ...
June 30, 2020 — A new study shows that SARS-CoV-2, the virus that causes COVID-19 (coronavirus), can infect heart cells ...
Change Healthcare Cardiology Hemodynamics is an integrated hemodynamic monitoring system for monitoring vital signs and ...
June 30, 2020 — BioCardia, Inc. announced that the company has resumed cases in the CardiAMP Heart Failure Trial. The ...
June 29, 2020 — Boston Scientific received U.S. Food and Drug Administration (FDA) 510(k) clearance for the LUX-Dx ...
June 29, 2020 — A type of smart magnetic resonance imaging (MRI) scan used in people with heart disease could help ...


 July 08, 2020
July 08, 2020














