July 1, 2020 — Carag AG announced receiving U.S. Food and Drug Administration (FDA) Investigational Device Exemption ...
In recent weeks, a multisystem hyperinflammatory condition has emerged in children in association with prior exposure or ...
June 30, 2020 — DiA Imaging Analysis, a leading provider of advanced AI-based solutions for ultrasound analysis ...
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
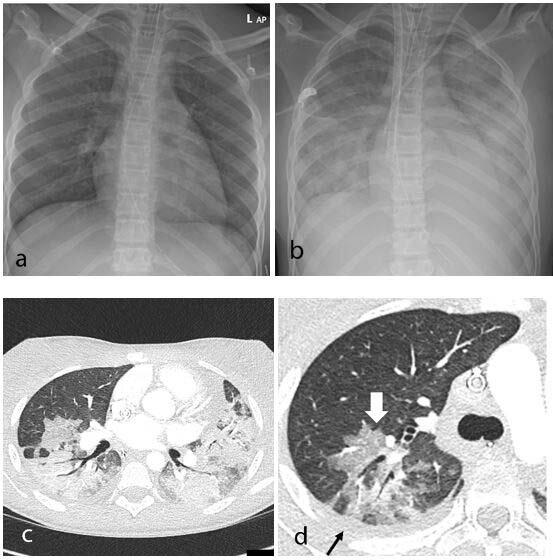
June 30, 2020 — A new study shows that SARS-CoV-2, the virus that causes COVID-19 (coronavirus), can infect heart cells ...
June 30, 2020 — BioCardia, Inc. announced that the company has resumed cases in the CardiAMP Heart Failure Trial. The ...
June 29, 2020 — Boston Scientific received U.S. Food and Drug Administration (FDA) 510(k) clearance for the LUX-Dx ...
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
June 29, 2020 — A type of smart magnetic resonance imaging (MRI) scan used in people with heart disease could help ...
June 26, 2020 — Abbott announced new data from the company's LightLab Initiative that showed optical coherence ...
June 26, 2020 – New data from the Global SYMPLICITY Registry (GSR) showed that renal denervation (RDN) with the ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
June 23, 2020 – Heart valve start-up Foldax is looking to reinventing several aspects of the prosthetic heart valve ...
June 23, 2020 — Philips announced the U.S. Food and Drug Administration (FDA) has granted premarket approval (PMA) for ...
June 23, 2020 — The U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Program status for the ...
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
Prior to January 2020 when clinicians read about the history of the 1918 flu, and epidemiologists predicted we were ...
June 19, 2020 — SMT (Sahajanand Medical Technologies Pvt. Ltd) said it acquired of the structural heart medical device ...
June 19, 2020 — iVascular SLU announced the global launch of Essential Pro, a novel coronary artery drug-coated balloon ...


 July 01, 2020
July 01, 2020