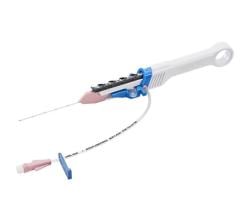
July 9. 2009 - Vascular Solutions, Inc. announces the availability of the new Trespass angiographic catheter specifically designed for use by interventional radiologists and other physicians performing endovascular Abdominal Aortic Aneurysm (AAA) repair procedures.
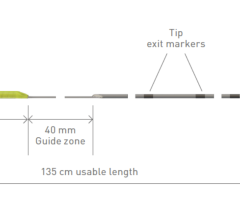
The catheter's hybrid design is said to offer simplicity and multiple functionality. The Trespass angiographic catheter combines an angled tip design to facilitate guidewire delivery and direction with radiopaque markers for vessel sizing and sideports for high pressure injections. Because the Trespass is a flush catheter, angled tip catheter and vessel sizing catheter all in one, it replaces multiple catheters that are commonly used in AAA procedures with just one. The catheter comes in a 5F, 65-cm configuration.
The Trespass catheter is intended for use for delivering radiopaque media to selected sites of the vascular system, and for pressure and anatomical measurements, and is currently available for sale in the United States.
For more information: www.vascularsolutions.com.


 January 29, 2026
January 29, 2026